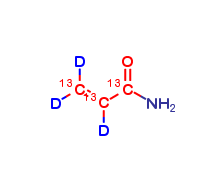
Acrylamide 13C3,2,3,3-D3
| Product Name | Acrylamide 13C3,2,3,3-D3 |
|---|---|
| Alternate Names | Acrylamide Stable Isotopes, Stable Isotopes of Acrylamide |
| CAT No. | CS-BX-00742 |
| CAS No. | 79-06-1 (Unlabeled) |
| Category | Stable Isotopes |
| Stock | Enquire |
| Mol. Wt. | 77.07 g/mol |
| Mol. For. | 13C3H2D3NO |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Acrylamide |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Acrylamide 13C3,2,3,3-D3 is a labeled form of acrylamide, a chemical compound with the molecular formula C3H5NO. It is a white crystalline solid that is soluble in water and has a melting point of 84.5°C. Acrylamide is widely used in industry for the production of polyacrylamide, a polymer that is used in various applications such as water treatment, papermaking, and oil recovery.
Acrylamide is also formed naturally in certain foods during high-temperature cooking processes such as frying, roasting, and baking. It is a known carcinogen and neurotoxin that has been linked to an increased risk of cancer and other health problems. The use of acrylamide in food production is regulated by various government agencies, and efforts are underway to reduce its presence in food products.
Acrylamide 13C3,2,3,3-D3 is a labeled form of acrylamide that is used in research and analytical applications to track the fate and metabolism of acrylamide in biological systems. It is also used in isotope labeling experiments to study the mechanisms of acrylamide formation in food and to develop new strategies for reducing its presence in food products. The labeled compound is synthesized using 13C and D isotopes, which replace some of the carbon and hydrogen atoms in the molecule, respectively. This allows researchers to distinguish the labeled compound from the unlabeled compound in biological samples and to track its movement and transformation in the body.
Get an Instant Quote
This page contains information about Acrylamide 13C3,2,3,3-D3. You can buy Acrylamide 13C3,2,3,3-D3 from Clearsynth at best competitive price with assured price guarantee. Clearsynth offers best quality Acrylamide 13C3,2,3,3-D3