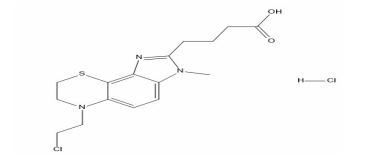
Phenylephrine Impurity D
| Product Name | Phenylephrine Impurity D |
|---|---|
| Alternate Names | Phenylephrine Impurities, Impurities of Phenylephrine |
| CAT No. | CS-DH-00008 |
| CAS No. | 1367567-95-0 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 257.33 g/mol |
| Mol. For. | C₁₆H₁₉NO₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Phenylephrine |
| Smileys | CN(CC1=CC=CC=C1)CC(C2=CC(=CC=C2)O)O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Phenylephrine Impurity D is a chemical compound that is used in the pharmaceutical industry as a reference standard for the analysis of pharmaceutical products. It is a known impurity that is present in some batches of Phenylephrine, which is a medication used for the treatment of nasal congestion, hypotension, and shock.
Chemically, Phenylephrine Impurity D is also known as (R)-3,4-dihydroxyphenylglycol. It is a chiral compound that is derived from the metabolism of norepinephrine, a neurotransmitter and hormone that plays a role in the body's response to stress.
Phenylephrine Impurity D is typically used as a reference standard in analytical methods such as high-performance liquid chromatography (HPLC) and gas chromatography (GC). These methods are used to analyze the purity and quality of Phenylephrine and other pharmaceutical products that contain this compound.
Phenylephrine Impurity D is also used in the development of new pharmaceutical products. By studying the properties and behavior of this impurity, researchers can gain insights into the metabolism and pharmacokinetics of Phenylephrine, which can lead to the development of more effective and safer medications.
In conclusion, Phenylephrine Impurity D is an important chemical compound in the pharmaceutical industry that is used as a reference standard for the analysis of pharmaceutical products. Its chemical properties and behavior provide valuable insights into the metabolism and pharmacokinetics of Phenylephrine and other related compounds, which can lead to the development of safer and more effective medications.
Get an Instant Quote
Related Compounds
Phenylephrine Impurity 20 | Phenylephrine Impurity C | Phenylephrine EP Impurity E | L-Phenylephrine sulfonate | Phenylephrine Impurity 9 | N-acetylphenylephrine | Phenylephrine Citrate Adduct -2 | Phenylephrine Impurity 33 | Phenylephrine Impurity 38 | N-Nitroso Phenylephrine (Mixture of Isomers) | Phenylephrine isoquinoline 4,6-diol analog | Phenylephrine isoquinolinone analog | Phenylephrine impurity 43 | Phenylephrine maleic acid adduct | rac 2-Bromo Phenylephrine Hydrochloride | rac Benzyl Phenylephrine | Phenylephrine Impurity 26 | Phenylephrine Impurity-39 | Phenylephrine EP Impurity C TFA salt | Phenylephrine Impurity 12 | Phenylephrine Impurity-41 | D-Phenylephrine sulfonate | N-(2-Succinyl) Phenylephrine | Phenylephrine Impurity 4 | Phenylephrine D-(+)-glucose Adduct | Phenylephrine EP Impurity C HCl | Phenylephrine Impurity 29 | O-Acetyl-(R)-phenylephrine Hydrochloride | Phenylephrine related compound F | Phenylephrine Impurity 5 | Phenylephrine Impurity-40 | Phenylephrine Impurity-37 | Phenylephrine EP Impurity A |