Afatinib 13C6
| Product Name | Afatinib 13C6 |
|---|---|
| Alternate Names | Afatinib Stable Isotopes, Stable Isotopes of Afatinib |
| CAT No. | CS-EK-02211 |
| CAS No. | 850140-72-6 unlabeled |
| Category | Stable Isotopes |
| Stock | Enquire |
| Mol. Wt. | Not Available |
| Mol. For. | Not Available |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Afatinib |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
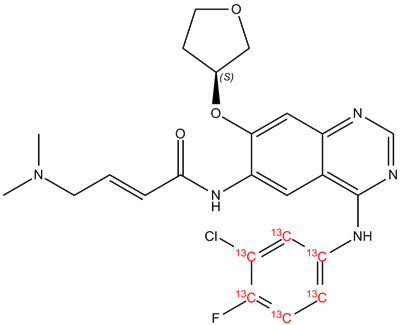
[13C6]-Afatinib is a stable isotope-labeled version of the drug Afatinib. It is commonly used in research studies to track the pharmacokinetics of Afatinib in vivo and in vitro. Afatinib is a tyrosine kinase inhibitor that selectively targets the epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), and HER4. This drug is used to treat non-small cell lung cancer (NSCLC) that has spread to other parts of the body.
The stable isotope-labeled version of Afatinib, [13C6]-Afatinib, contains six carbon atoms that are replaced with the stable isotope carbon-13. This labeling technique allows researchers to track the drug's metabolism in the body and determine its pharmacokinetics. Moreover, it is a powerful tool in the study of drug-drug interactions and drug metabolism.
In addition to its use in research studies, [13C6]-Afatinib has the same pharmacological properties as Afatinib and is used to treat patients with NSCLC. However, it is not commonly used in clinical settings due to its high cost and limited availability.
Overall, [13C6]-Afatinib is an essential tool in the study of Afatinib's pharmacokinetics and drug interactions. It provides valuable insights that help to improve the efficacy and safety of Afatinib and other tyrosine kinase inhibitors in the treatment of NSCLC.
Get an Instant Quote
Related Compounds
Afatinib D4 | Afatinib D6 |