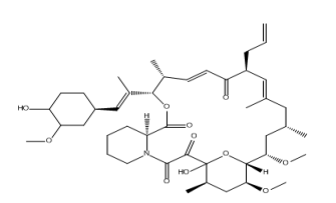
Tacrolimus Dehydro Impurity
| Product Name | Tacrolimus Dehydro Impurity |
|---|---|
| Alternate Names | Tacrolimus Impurities, Impurities of Tacrolimus |
| CAT No. | CS-EO-00669 |
| CAS No. | 129212-37-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 786.00 g/mol |
| Mol. For. | C₄₄H₆₇NO₁₁ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Tacrolimus |
| Smileys | O[C@H]1[C@H](OC)C[C@H](/C=C(C)/[C@@H](OC([C@@H]2CCCCN2C(C([C@]3(O)O[C@H]([C@@H](OC)C4)[C@@H](OC)C[C@H]3C)=O)=O)=O)[C@@H](/C=C/C([C@@H](CC=C)/C=C(C)/C[C@@H]4C)=O)C)CC1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Tacrolimus Dehydro Impurity is a substance that is commonly used in the pharmaceutical industry as an intermediate in the synthesis of Tacrolimus, a potent immunosuppressive drug used to prevent organ rejection in transplant patients. The Dehydro Impurity of Tacrolimus is a by-product that is formed during the manufacturing process of Tacrolimus and is usually present in small amounts.
Chemically, Tacrolimus Dehydro Impurity is a derivative of Tacrolimus and has a similar structure to it. It is a colorless to pale yellow solid that is sparingly soluble in water and soluble in organic solvents like methanol, ethanol, and acetone. Its molecular formula is C44H65NO13 and its molecular weight is 825.98 g/mol.
Tacrolimus Dehydro Impurity is mainly used as a reference standard in the quality control of Tacrolimus. It is also used in the development and validation of analytical methods for the determination of Tacrolimus and its related substances. Additionally, it is used in the research of the structure-activity relationship of Tacrolimus derivatives.
In conclusion, Tacrolimus Dehydro Impurity plays an important role in the pharmaceutical industry, particularly in the quality control and research of Tacrolimus. Its chemical properties and structure make it a valuable reference standard for the development and validation of analytical methods in the determination of Tacrolimus and its related substances.
Get an Instant Quote
Related Compounds
Tacrolimus Impurity I | Tacrolimus Methyl Acryl Aldehyde | Tacrolimus alpha-Hydroxy Acid (TAC-H1) | Tacrolimus EP Impurity H | Tacrolimus EP Impurity B | Tacrolimus 21-carboxylic acid | Tacrolimus Impurity 6 (Tacrolimus Hydroxy Acid Impurity) | Tacrolimus Lactone Isomer | Tacrolimus Impurity 18 | Tacrolimus Impurity 21 | Tacrolimus Impurity 23 | Tacrolimus 8-Propyl Analog | Tacrolimus impurity | Tacrolimus Related Compound A | Tacrolimus EP Impurity F | Tacrolimus Impurity X | Tacrolimus Diene | Tacrolimus Ring-opening Impurity | Oxacyclohexane Open Ring Tacrolimus | Tacrolimus C4-epimer Diene | Tacrolimus impurity-1 | Tacrolimus Butenyl analogue | Tacrolimus Impurity 39 | Tacrolimus Impurity III | Tacrolimus Regioisomer | Tacrolimus Impurity VII |