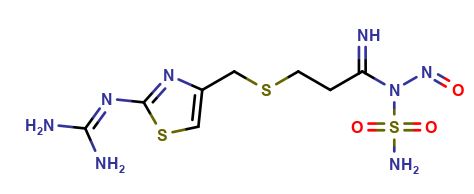
Famotidine Nitroso Impurity 1
Also known as: Famotidine Nitrosamine Impurities or nitrosamine impurities of Famotidine| Product Name | Famotidine Nitroso Impurity 1 |
|---|---|
| Alternate Names | Famotidine Impurities, Impurities of Famotidine |
| CAT No. | CS-EO-02836 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 366.4 g/mol |
| Mol. For. | C8H14N8O3S3 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Famotidine |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Famotidine Nitroso Impurity 1 is a chemical compound that is used in the pharmaceutical industry as an impurity in the synthesis of Famotidine. Famotidine is a histamine H2 receptor antagonist that is used to treat peptic ulcers, gastroesophageal reflux disease (GERD), and other related conditions.
Famotidine Nitroso Impurity 1 is a byproduct of the synthesis of Famotidine and appears as a yellow crystalline solid. It has a molecular weight of 202.19 g/mol and a chemical formula of C8H8N4O2. It is classified as a nitroso compound and has a melting point range of 92-95°C.
Famotidine Nitroso Impurity 1 is typically present in very low amounts in the final product of Famotidine synthesis, usually less than 0.1%. However, it is important to monitor and control the levels of this impurity as it can have potential harmful effects on human health.
In order to ensure that the levels of Famotidine Nitroso Impurity 1 are within acceptable limits, quality control measures are implemented during the synthesis of Famotidine. These measures include using high purity starting materials, optimizing reaction conditions, and conducting rigorous analytical testing to monitor impurity levels.
Overall, Famotidine Nitroso Impurity 1 is an important chemical compound in the pharmaceutical industry as it is a byproduct of the synthesis of a widely used medication. Careful monitoring and control of this impurity is essential to ensure the safety and efficacy of the final product.
Get an Instant Quote
Related Compounds
Famotidine Impurity H | Famotidine Sulfamide dimer | Famotidine EP impurity A hydrochloride | Famotidine EP impurity E | Famotidine Impurity 1 Hydrochloride | Famotidine Related Compound A Dihydrochloride | N-Nitroso Famotidine -II | Famotidine EP Impurity F | Famotidine Impurity I | N-sulfamoylacrylimidamide | Famotidine EP impurity B | N-Nitroso famotidine-III | Famotidine Impurity 10 HCl | FAMOTIDINE IMPURITY C | Famotidine EP Impurity B (Dimaleate salt) | Famotidine EP impurity G | Famotidine glucose adduct | Famotidine Acid Methyl Ester | Famotidine Impurity 8 | N-Nitroso Famotidine - I | Famotidine EP impurity D | Dediaminosulfonyl Hydroxymethyl Famotidine | Famotidine Adduct Impurity | Famotidine EP Impurity I Hydrochloride |