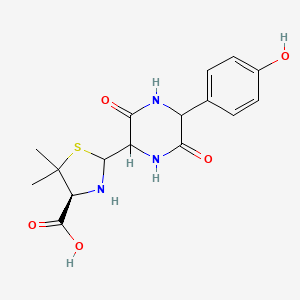
Amoxicillin EP Impurity C
| Product Name | Amoxicillin EP Impurity C |
|---|---|
| Alternate Names | Amoxicillin Impurities, Impurities of Amoxicillin |
| CAT No. | CS-O-07189 |
| CAS No. | 2088961-37-7 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 365.4 g/mol |
| Mol. For. | C₁₆H₁₉N₃O₅S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Amoxicillin |
| Purity | Not less than 95 % |
| Therapeutic | Anti ulcer |
| Smileys | CC1(C(NC(S1)C2C(=O)NC(C(=O)N2)C3=CC=C(C=C3)O)C(=O)O)C |
| Canonical Smiles | CC1(C(NC(S1)C2C(=O)NC(C(=O)N2)C3=CC=C(C=C3)O)C(=O)O)C |
| InchIKey | IIZCCQJEPBWGJU-DGVZPSOQSA-N |
| Inchl | InChI=1S/C16H19N3O5S/c1-16(2)11(15(23)24)19-14(25-16)10-13(22)17-9(12(21)18-10)7-3-5-8(20)6-4-7/h3-6,9-11,14,19-20H,1-2H3,(H,17,22)(H,18,21)(H,23,24)/t9?,10?,11-,14?/m0/s1 |
| IUPAC | (4S)-2-[5-(4-hydroxyphenyl)-3,6-dioxopiperazin-2-yl]-5,5-dimethyl-1,3-thiazolidine-4-carboxylic acid |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Amoxicillin EP Impurity C, also known as 2-Hydroxyethylthioethyl amoxicillin, is an organic compound that is used in the pharmaceutical industry as an impurity reference standard for the analysis of amoxicillin drug substances and products. The impurity is derived from the synthesis of amoxicillin, which is a β-lactam antibiotic used to treat bacterial infections. The presence of Amoxicillin EP Impurity C in amoxicillin drug substances and products can affect their quality and efficacy, and therefore, its quantification is important for ensuring the safety and effectiveness of amoxicillin-based medications.
Chemically, Amoxicillin EP Impurity C is a white to off-white crystalline powder with a molecular formula of C11H20N2O5S and a molecular weight of 288.35 g/mol. It is soluble in water and ethanol, and its melting point ranges from 98°C to 102°C. The impurity is structurally related to amoxicillin, containing a β-lactam ring and an amide group.
The usage of Amoxicillin EP Impurity C involves its quantification in amoxicillin drug substances and products using various analytical techniques such as high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS). The impurity reference standard is used to establish the limit for its acceptable levels in amoxicillin-based medications, which is typically set at 0.2% or lower.
In conclusion, Amoxicillin EP Impurity C is an important reference standard used in the pharmaceutical industry for the analysis of amoxicillin drug substances and products. Its quantification is crucial for ensuring the safety and effectiveness of amoxicillin-based medications, and its chemical information and properties are important for its identification and characterization.
Get an Instant Quote
Related Compounds
Amoxicillin Impurity C (R Isomer) | Amoxicillin EP Impurity L | Amoxicillin EP Impurity O | N-Nitroso Amoxicillin | Amoxicillin EP Impurity J | Amoxicillin Close ring Trimer | Amoxicillin EP Impurity P | Amoxicillin Open Ring Ethyl | Amoxicillin Trimer Trisodium Salt | Amoxicilloic Acid | Amoxicillin Related Compound M | Amoxicillin EP Impurity H | Amoxicillin Related Compound D | p-hydroxyphenylglycine methyl ester | Amoxicillin Related Compound E | Amoxicillin EP Impurity H | Amoxicilline EP Impurity D (Mixture of isomers) | Amoxicillin Aldehyde | Amoxicillin EP Impurity K | Amoxicillin Open Ring Trimer Impurity | Amoxicillin EP Impurity J Disodium salt | Amoxicillin oxide | Amoxicillin Open Ring Decarboxylated Dimer | Amoxicillin EP Impurity F | rac-Amoxicillin EP Impurity H | N-Pivaloyl Amoxicillin | Sterile Clavulanate Potassium+Amoxicillin | Amoxicillin Trihydrate - Impurity K | Amoxicillin Open RIng Trimer Impurity sodium salt | Amoxicillin EP Impurity B | Amoxicillin double Side Chain Impurity | N-(Hydroxyphenylglycyl) amoxicillin Sodium salt | Amoxicillin EP Impurity I | Amoxicillin EP Impurity K trisodium salt | Amoxicillin Impurity M | Amoxicillin Dimer (Penicilloic acid form) | Amoxicillin Related Compound C | Amoxicillin Related Compound M Sodum salt | Amoxicillin Trihydrate - Impurity J | Amoxicillin Trimer |