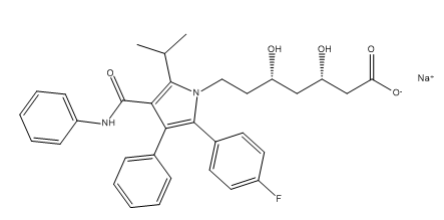
Atorvastatin EP Impurity E Sodium salt
| Product Name | Atorvastatin EP Impurity E Sodium salt |
|---|---|
| Alternate Names | Atorvastatin Impurities, Impurities of Atorvastatin |
| CAT No. | CS-O-07289 |
| CAS No. | 501121-34-2 (Free Acid) |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 580.62 g/mol |
| Mol. For. | C₃₃H₃₄FN₂NaO₅ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Atorvastatin |
| Therapeutic | Anti-Diabetic |
| Smileys | O=C(C(C(C1=CC=CC=C1)=C(C2=CC=C(F)C=C2)N3CC[C@H](O)C[C@H](O)CC([O-])=O)=C3C(C)C)NC4=CC=CC=C4.[Na+] |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Atorvastatin EP Impurity E, also known as Methyl 4-(4-fluorophenyl)-6-isopropyl-2-[(N-methylmethanesulfonamido)methyl]pyridine-3-carboxylate, is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for quality control purposes. This impurity is typically found in small amounts in Atorvastatin, which is a popular drug used to lower cholesterol levels in patients with hypercholesterolemia.
Atorvastatin EP Impurity E is a pyridine-based compound that contains a fluorophenyl group, a methyl group, and an isopropyl group. The compound has a molecular formula of C22H28FN2O4S and a molecular weight of 442.54 g/mol. It is a white to off-white powder that is sparingly soluble in water but highly soluble in organic solvents like methanol, ethanol, and acetone.
In pharmaceutical applications, Atorvastatin EP Impurity E is used as a reference standard in the development and validation of analytical methods for the quantification of impurities in Atorvastatin. It is also used in the identification and characterization of impurities that may be present in Atorvastatin samples. The use of reference standards like Atorvastatin EP Impurity E ensures that pharmaceutical products are of consistent quality and purity, which is essential for patient safety and efficacy.
Get an Instant Quote
Related Compounds
Atorvastatin 2-Fluoro t-Butyl Ester | Atorvastatin Cyclic (Fluorophenyl) Sodium | Atorvastatin EP Impurity B | Atorvastatin epoxy pyrrolooxazin 7-hydroxy analog | Allyl Ester of Atorvastatin Cyclic (Isopropyl) Impurity | Atorvastatin Impurity 1 | Atorvastatin Di-acetonide tert-Butyl Ester | sodium (3R,5R)-7-(2-(4-fluorophenyl)-5-isopropyl-3-phenyl-1H-pyrrol-1-yl)-3,5-dihydroxyheptanoate | Atorvastatin Pyrrolidone Analog | Atorvastatin Ethyl Ester | Atorvastatin Diketoene Impurity | Atorvastatin EP Impurity F (Ca Salt) | Atorvastatin EP Impurity B Calcium salt | Atorvastatin Impurity 16 | Atorvastatin EP Impurity C Sodium salt | Atorvastatin Impurity 25 | Atorvastatin Dehydro Sodium Salt (E/Z mixture) | Atorvastatin 3-Deoxyhept-2Z-Enoic Acid Sodium Salt | Atorvastatin EP Impurity Q sodium salt | Allyl Ester of Atorvastatin Cyclic (Fluorophenyl) Impurity | Atorvastatin-ATN-2 triamino impurity | Atorvastatin Lactam Impurity calcium salt | N-Nitroso Atorvastatin Impurity | Atorvastatin Impurity F | Atorvastatin EP Impurity P (Sodium salt) | Atorvastatin Acid Methyl Ester | Atorvastatin Impurity 19 | Atorvastatin Pyrrolidone Phenanthrene Sodium | Atorvastatin Calcium Propylene Glycol Solvate | Atorvastatin 3-Deoxyhept-2E-Enoic Acid | Atorvastatin Amide | Atorvastatin EP Impurity A (Sodium Salt) | Atorvastatin Allyl Ester | Atorvastatin Lactam Calcium Salt Impurity | Atorvastatin 3-Deoxy-Hept-2-Enoic Acid Ethyl Ester | Atorvastatin EP Impurity O Sodium salt | Atorvastatin 3-Fluoro Analog | Atorvastatin EP Impurity G (3-O-methyl Atorvastatin calcium salt) | Atorvastatin Dehydro Lactone | Atorvastatin Impurity 21 | Atorvastatin Cyclic Fluorophenyl calcium Salt Impurity | Atorvastatin Pyrrolidone Impurity | Atorvastatin Acid Sodium salt | Difluoro Atorvastatin Acetonide tert-Butyl Ester | Atorvastatin diacid impurity | Atorvastatin 2-Fluoro Analog | Dihydroxy Diketo Atorvastatin Impurity | Atorvastatin Cyclic (Fluorophenyl) Impurity | Atorvastatin Diepoxide Impurity | Atorvastatin Impurity 20 | Atorvastatin EP Impurity C | Atorvastatin EP Impurity D | Atorvastatin Lactone 3-O-Methyl Ether | Atorvastatin Diamino impurity | Atorvastatin Epoxy Pyrrolooxazin Analog | Atorvastatin Dehydro Acid Sodium Salt | ATV-XI | Atorvastatin EP Impurity A (Sodium salt) | Atorvastatin Lactam Phenanthrene | Atorvastatin Epoxy Tetrahydrofuran Analog | Atorvastatin Impurity 17 | Atorvastatin EP Impurity I | Atorvastatin Cyclic (Fluorophenyl) Impurity | Atorvastatin EP Impurity A | Atorvastatin Acetonide Methyl Ester | Atorvastatin Lactam Sodium Salt Impurity | Atorvastatin Epoxy Pyrrolooxazin tricyclic Calcium Salt | Atorvastatin Pyrrolidone Phenanthrene Calcium | Difluoro Atorvastatin | Atorvastatin Impurity D |