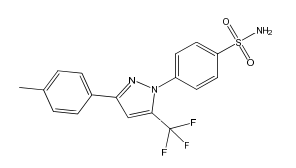
Celecoxib EP Impurity B
| Product Name | Celecoxib EP Impurity B |
|---|---|
| Alternate Names | Celecoxib Impurities, Impurities of Celecoxib |
| CAT No. | CS-O-07592 |
| CAS No. | 331943-04-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 381.37 g/mol |
| Mol. For. | C₁₇H₁₄F₃N₃O₂S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Celecoxib |
| Purity | 95% |
| Smileys | CC1=CC=C(C=C1)C2=NN(C(=C2)C(F)(F)F)C3=CC=C(C=C3)S(=O)(=O)N |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Celecoxib EP Impurity B is a chemical compound that is used in the pharmaceutical industry as an impurity reference standard for the analysis of Celecoxib, a nonsteroidal anti-inflammatory drug. Celecoxib is mainly used to treat pain and inflammation associated with various conditions such as arthritis, menstrual cramps, and acute pain. Celecoxib EP Impurity B is used to ensure the quality and purity of Celecoxib by acting as a reference standard for the analysis of impurities that may be present in the drug substance.
Chemically, Celecoxib EP Impurity B is known as 4-[4-(methylsulfonyl)phenyl]-2-(trifluoromethyl)benzoic acid. It is a white to off-white powder with a purity of not less than 98%. The chemical formula of Celecoxib EP Impurity B is C15H11F3O4S, and its molecular weight is 363.3 g/mol.
The usage of Celecoxib EP Impurity B involves its incorporation into the analytical methods used for the determination of impurities in Celecoxib. It is essential to ensure that the levels of impurities in Celecoxib are within acceptable limits to ensure the safety and efficacy of the drug. Celecoxib EP Impurity B is also used as a reference standard for the development and validation of analytical methods for Celecoxib, which is crucial in ensuring the accuracy and precision of the analytical results.
In conclusion, Celecoxib EP Impurity B is a critical reference standard used in the analysis of Celecoxib. It plays a vital role in ensuring the quality and purity of Celecoxib, a widely used drug for pain and inflammation. Its usage in the pharmaceutical industry is essential in ensuring the safety and efficacy of the drug.
Get an Instant Quote
Related Compounds
Celecoxib Impurity 23 | Celecoxib Impurity 4 | Celecoxib Impurity 7 | Celecoxib Impurity 21 | N-NITROSOETHYLENE DIAMINO TRIACETIC ACID | Celecoxib impurity 1 | Celecoxib Impurity 8 | Celecoxib Trifluro Impurity | Celecoxib Impurity 22 | Celecoxib Impurity 3 | Celecoxib nitroso impurity | Celecoxib Impurity 2 | Celecoxib EP Impurity A | N,N-DINITROSO-ETHYLENE DIAMINODIACETIC ACID |