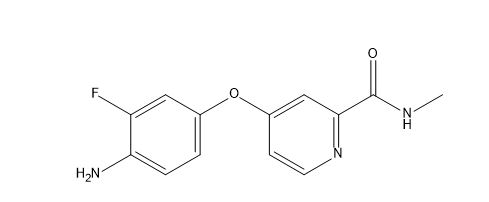
AFP-Picolin Methylamide Impurity
| Product Name | AFP-Picolin Methylamide Impurity |
|---|---|
| Alternate Names | Regorafenib Impurities, Impurities of Regorafenib |
| CAT No. | CS-O-11141 |
| CAS No. | 757251-39-1 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 261.25 g/mol |
| Mol. For. | C₁₃H₁₂FN₃O₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Regorafenib |
| Purity | >98% |
| Therapeutic | Anti-Cancer / Oncology |
| Smileys | FC1=C(N)C=CC(OC2=CC=NC(C(NC)=O)=C2)=C1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
AFP-Picolin Methylamide Impurity is a chemical compound used in the pharmaceutical industry as a reference standard in analytical testing. It is also used in the development and validation of analytical methods for quality control of pharmaceuticals. The compound is a known impurity of AFP-Picolin, which is a potential drug candidate for the treatment of cancer.
Chemically, AFP-Picolin Methylamide Impurity is a derivative of AFP-Picolin, which is a type of pyridine-based molecule. The compound is characterized by the presence of a methylamide group, which is attached to the pyridine ring. The impurity is formed during the synthesis of AFP-Picolin and can significantly affect the purity and quality of the final drug product.
Analytical methods such as High-Performance Liquid Chromatography (HPLC) and Gas Chromatography-Mass Spectrometry (GC-MS) are used to detect and quantify AFP-Picolin Methylamide Impurity in pharmaceutical samples. The impurity is considered harmful to human health and must be controlled within acceptable limits during drug development and manufacturing.
In conclusion, AFP-Picolin Methylamide Impurity is a critical compound in the pharmaceutical industry, used for analytical testing and quality control of potential cancer drugs. Its chemical properties and analytical methods used for its detection and quantification are essential for ensuring the safety and efficacy of pharmaceutical products.
Get an Instant Quote
Related Compounds
Regorafenib Impurity 4 | Regorafenib Impurity 1 | Regorafenib Urea Impurity | N-Desmethyl N-Hydroxymethyl Regorafenib | Regorafenib EP Impurity D | Regorafenib acid Impurity | Regorafenib Ethyl Carbamate Impurity | Regorafenib Impurity 3 | Regorafenib Dipyrimidine Impurity | Regorafenib Impurity 2 |