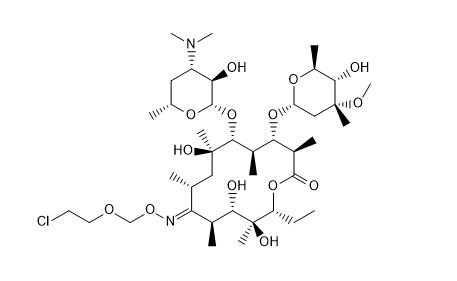
Roxithromycin EP Impurity J
| Product Name | Roxithromycin EP Impurity J |
|---|---|
| Alternate Names | Roxithromycin Impurities, Impurities of Roxithromycin |
| CAT No. | CS-O-11803 |
| CAS No. | 134834-12-1 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 841.47 g/mol |
| Mol. For. | C₄₀H₇₃ClN₂O₁₄ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Roxithromycin |
| Purity | 95% |
| Therapeutic | Antibiotics |
| Smileys | C[C@@H]([C@]([C@H](C(O[C@@H]1CC)=O)C)([H])O[C@@](O[C@@H](C)[C@@H]2O)([H])C[C@@]2(C)OC)[C@H]([C@](O)(C[C@H](/C([C@@H]([C@@H](O)[C@@]1(O)C)C)=N\OCOCCCl)C)C)O[C@@](O[C@H](C)C[C@@H]3N(C)C)([H])[C@@H]3O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Roxithromycin EP Impurity J is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard or impurity in the quality control of Roxithromycin, an antibiotic medication used to treat various bacterial infections. It is also known as 9-desoxo-9a-methyl-9a-aza-9a-homoerythromycin A-6, and its molecular formula is C40H69N3O13.
In terms of its chemical properties, Roxithromycin EP Impurity J is a semi-synthetic derivative of erythromycin, which belongs to the macrolide family of antibiotics. It has a complex structure consisting of a 14-membered lactone ring, a sugar moiety, and an amino acid side chain. The presence of a nitrogen atom in the lactone ring makes it aza-macrolide, which is more stable and less prone to microbial resistance than other macrolides.
The usage of Roxithromycin EP Impurity J is mainly for analytical purposes, such as identification, quantification, and characterization of Roxithromycin and its related substances in drug products or raw materials. It can be used as a chromatographic standard in high-performance liquid chromatography (HPLC) or gas chromatography (GC), or as a reference material in spectroscopic techniques such as nuclear magnetic resonance (NMR) or mass spectrometry (MS).
In conclusion, Roxithromycin EP Impurity J is a valuable chemical compound that plays a crucial role in ensuring the quality and safety of Roxithromycin-based medications. Its chemical information and usage are essential for pharmaceutical companies, regulatory agencies, and researchers who work in the field of antibiotics and drug development.
Get an Instant Quote
Related Compounds
Roxithromycin EP Impurity B | Roxithromycin EP Impurity E | Roxithromycin EP Impurity K | Roxithromycin EP impurity G | Roxithromycin EP Impurity C | Roxithromycin EP Impurity H | N-Nitroso N-Demethyl Roxithromycin | Roxithromycin EP Impurity F | Roxithromycin EP Impurity I | Roxithromycin EP Impurity D |