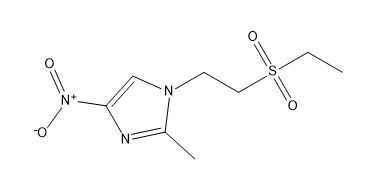
Tinidazole EP Impurity B
| Product Name | Tinidazole EP Impurity B |
|---|---|
| Alternate Names | Tinidazole Impurities, Impurities of Tinidazole |
| CAT No. | CS-O-13368 |
| CAS No. | 25459-12-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 247.27 g/mol |
| Mol. For. | C₈H₁₃N₃O₄S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Tinidazole |
| Purity | 95% |
| Smileys | CCS(=O)(=O)CCN1C=C(N=C1C)[N+](=O)[O-] |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Tinidazole EP Impurity B, also known as 1-(2-Ethylsulfanyl-1H-imidazol-5-yl)ethanone, is a chemical compound that is used in the pharmaceutical industry as a reference standard and impurity. It is a yellow to brown crystalline substance that is soluble in organic solvents like methanol and acetonitrile.
Tinidazole is a medication used to treat infections caused by certain bacteria and parasites. The presence of impurities in Tinidazole can affect its safety and efficacy. Therefore, it is important to monitor and control the levels of impurities in the drug during the manufacturing process. Tinidazole EP Impurity B is one of the impurities that can be present in the drug, and its levels need to be regulated to ensure the safety and effectiveness of the medication.
The chemical information of Tinidazole EP Impurity B includes its molecular formula, which is C7H10N2OS, and its molecular weight, which is 174.23 g/mol. It is also known as 2-Ethylsulfanyl-5-imidazolylacetone or Ethyl 2-(2-ethylsulfanyl-1H-imidazol-5-yl)acetate. The compound can be synthesized by several methods, including the reaction of ethyl 2-bromoacetate with 2-mercaptoimidazole, followed by hydrolysis and decarboxylation.
In conclusion, Tinidazole EP Impurity B is an important chemical compound used in the pharmaceutical industry as a reference standard and impurity. Its presence in Tinidazole medication needs to be monitored and controlled to ensure the safety and effectiveness of the drug. The chemical information of Tinidazole EP Impurity B includes its molecular formula, molecular weight, and synthesis methods.
Get an Instant Quote
This page contains information about Tinidazole EP Impurity B. You can buy Tinidazole EP Impurity B from Clearsynth at best competitive price with assured price guarantee. Clearsynth offers best quality Tinidazole EP Impurity B