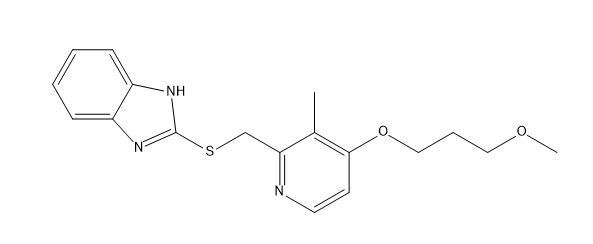
Rabeprazole Impurity F
| Product Name | Rabeprazole Impurity F |
|---|---|
| Alternate Names | Rabeprazole Impurities, Impurities of Rabeprazole |
| CAT No. | CS-O-13748 |
| CAS No. | 117977-21-6 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 343.44 g/mol |
| Mol. For. | C₁₈H₂₁N₃O₂S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Rabeprazole |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Rabeprazole Impurity F is a chemical compound that is used in the pharmaceutical industry as a reference standard or impurity marker for the quality control of rabeprazole. Rabeprazole is a proton pump inhibitor that is used in the treatment of gastroesophageal reflux disease (GERD), peptic ulcers, and other acid-related disorders. Rabeprazole Impurity F is one of the impurities that can be present in rabeprazole samples, and its presence can affect the efficacy and safety of the drug.
Chemically, Rabeprazole Impurity F is known as 1-(3-methoxypropyl)-2-(4-methoxy-3-methylpyridin-2-yl) benzimidazole. Its molecular formula is C18H22N4O2, and its molecular weight is 330.4 g/mol. Rabeprazole Impurity F is a white to off-white crystalline powder that is sparingly soluble in water but soluble in most organic solvents.
The chemical information of Rabeprazole Impurity F is important for the development and manufacture of rabeprazole. It is used as a reference standard to determine the purity and identity of rabeprazole samples. This impurity can be detected and quantified using analytical techniques such as high-performance liquid chromatography (HPLC) and gas chromatography (GC). By ensuring that rabeprazole samples meet the required quality standards, the safety and efficacy of the drug can be guaranteed.
Get an Instant Quote
Related Compounds
Rabeprazole Sodium hydrate | Rabeprazole Impurity (Chloro Intermediate) | Nitroso Rabeprazole | Rabeprazole Impurity A | Rabeprazole Impurity D Sulfide | Rabeprazole Sodium Hydrate | Rabeprazole USP Related Compound A | Rabeprazole Impurity 11 | Rabeprazole EP Impurity C | Rabeprazole 6-chloro Sodium salt | Rabeprazole 5 -chloro Impurity | Rabeprazole Impurity G Sulfone | Rabeprazole N-alkylated Sulphide Impurity | Rabeprazole Carboxylic Acid Impurity | Rabeprazole EP Impurity I | Rabeprazole Impurity G | Rabeprazole 2-Chloromethyl Impurity | Rabeprazole picolinium salt |