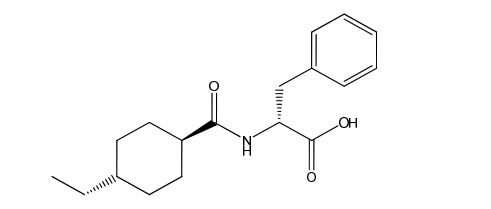
Nateglinide EP Impurity E
| Product Name | Nateglinide EP Impurity E |
|---|---|
| Alternate Names | Nateglinide Impurities, Impurities of Nateglinide |
| CAT No. | CS-O-13902 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 303.40 g/mol |
| Mol. For. | C₁₈H₂₅NO₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Nateglinide |
| Purity | 95% |
| Therapeutic | Anti-Diabetic |
| Smileys | O=C([C@H]1CC[C@H](CC)CC1)N[C@@H](C(O)=O)CC2=CC=CC=C2 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Nateglinide EP Impurity E is a chemical compound that is used in the pharmaceutical industry to produce drugs used to treat diabetes. It is a by-product of the synthesis of nateglinide, a medication that belongs to the class of drugs known as meglitinides. Nateglinide EP Impurity E is classified as an impurity because it is formed during the manufacturing process of nateglinide and is not intended to be present in the final product.
Chemically, Nateglinide EP Impurity E is a complex organic compound that contains a heterocyclic ring structure. It is soluble in organic solvents like methanol, ethanol, and acetonitrile. The exact chemical composition of Nateglinide EP Impurity E is not publicly disclosed due to proprietary information of the manufacturers.
The presence of impurities in pharmaceutical products is strictly regulated by various regulatory agencies like the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Therefore, the amount of Nateglinide EP Impurity E present in nateglinide is closely monitored and controlled to ensure that it does not exceed the allowable limits.
In conclusion, Nateglinide EP Impurity E is a chemical compound that is important in the production of nateglinide, a medication that is used to treat diabetes. Its chemical properties and exact composition are not publicly disclosed, and its presence in the final product is closely monitored by regulatory agencies to ensure that it does not exceed allowable limits.
Get an Instant Quote
Related Compounds
Nateglinide Impurity A | ent-Nateglinide | Nateglinide Methyl Ester | Nateglinide EP Impurity G | Nateglinide EP Impurity F [Mixture of isomers] |