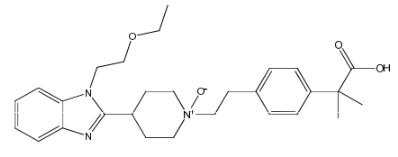
Bilastine N-oxide Impurity
| Product Name | Bilastine N-oxide Impurity |
|---|---|
| Alternate Names | Bilastine Impurities, Impurities of Bilastine |
| CAT No. | CS-O-14025 |
| CAS No. | 2069238-47-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 479.61 g/mol |
| Mol. For. | C₂₈H₃₇N₃O₄ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Bilastine |
| Therapeutic | Antihistamine |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Bilastine N-oxide Impurity is a chemical compound that is commonly used in the pharmaceutical industry. It is a byproduct of the synthesis of bilastine, which is an antihistamine drug used to treat various allergic conditions. The impurity is formed during the manufacturing process of bilastine, and it is typically removed from the final product through purification and filtration techniques.
The usage of Bilastine N-oxide Impurity is limited to research and development purposes. It is not intended for human consumption or therapeutic use, as its effects on the body are not fully understood. However, this impurity serves as an important marker for quality control and purity testing of bilastine. It is also used in analytical methods to assess the stability and degradation of bilastine over time.
Chemically, Bilastine N-oxide Impurity is a derivative of bilastine, with an additional oxygen molecule attached to its structure. Its molecular weight is slightly higher than that of bilastine, and it has a distinct chemical profile that can be identified through spectroscopic techniques. The impurity is considered to be a minor component in the final product, and its presence is closely monitored to ensure the safety and effectiveness of bilastine as a drug.
Get an Instant Quote
Related Compounds
Desethoxyethyl Bilastine | Methyl 2-(4-(2-chloroethyl)phenyl)-2-methylpropanoate | trans-Bilastine N-Oxide | Bilastine Nitroso impurity-3 | Bilastine Oxo impurity | Bilastine Impurity 3 | Methyl 2-(4-(2-chloroacetyl) phenyl)-2-methyl propanoate | Bilastine Impurity 28 | Desethoxy Ethyl Bilastine Impurity | Bilastine Impurity 10 | Bilastine Impurity 1 | Bilastine Methyl Ester | Bilastine Nitroso Impurity 2 | Bilastine Related Compound C | Methyl 2-(4-(2-bromoacetyl)phenyl)-2-methylpropanoate | Bilastine Impurity B | Bilastine Hydroxy Impurity | Bilastine Impurity 20 | Bilastine Dimethylamide impurity | Bilastine Propanamide Impurity | Bilastine Impurity C | Bilastine Impurity 7 | Bilastine Chloro Impurity | Bilastine Keto Impurity | Bilastine-N-Nitrosoamine Imputrity -1 | Bilastine Impurity 13 | cis-Bilastine N-Oxide | Bilastine Impurity 9 | Bilastine Boc Impurity | Bilastine Impurity A |