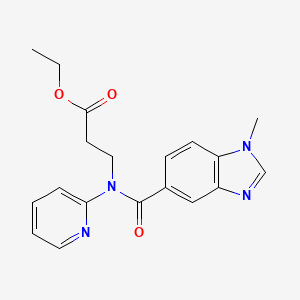
Dabigatran Impurity 2
| Product Name | Dabigatran Impurity 2 |
|---|---|
| Alternate Names | Dabigatran Impurities, Impurities of Dabigatran |
| CAT No. | CS-O-14955 |
| CAS No. | 1702936-92-2 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 352.39 g/mol |
| Mol. For. | C₁₉H₂₀N₄O₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Dabigatran |
| Therapeutic | Anti-Thrombotics |
| Smileys | CN1C2=CC=C(C=C2N=C1)C(N(CCC(OCC)=O)C3=CC=CC=N3)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Dabigatran Impurity 2 is a chemical compound that is commonly used as an impurity reference standard in the manufacturing of dabigatran etexilate, a direct thrombin inhibitor used in the treatment of thromboembolic diseases such as deep vein thrombosis and pulmonary embolism. This impurity is a byproduct of the synthesis of dabigatran etexilate, and its presence in the final product can affect the efficacy and safety of the medication.
The chemical name of Dabigatran Impurity 2 is N-(2-hydroxy-1,1-dimethylethyl)-3-[(2-methyl-1H-imidazol-1-yl)methyl]-2-thioxo-2,3-dihydro-1H-pyrido[3,4-b]indole-1-carboxamide. Its molecular formula is C21H24N6O2S, and its molecular weight is 440.52 g/mol.
The usage of Dabigatran Impurity 2 is strictly regulated by the FDA and other regulatory bodies to ensure the quality and safety of dabigatran etexilate. It is typically used as a reference standard in the quality control testing of dabigatran etexilate to ensure that the impurity levels are within acceptable limits. The impurity is also used in research and development of new formulations of dabigatran etexilate.
Overall, Dabigatran Impurity 2 is an important chemical compound in the manufacturing of dabigatran etexilate and plays a critical role in ensuring the efficacy and safety of this medication.
Get an Instant Quote
Related Compounds
Dabigatran Impurity K | ethyl 3-(2-(((4-(((hexyloxy)carbonyl)carbamoyl)phenyl)amino)methyl)-1-methyl-N-(pyridin-2-yl)-1H-ben | N-Nitroso Dabigatran Etexilate | Dabigatran Impurity 28 | Dabigatran Etexilate Impurity 1 | Dabigatran Ethyl Ester Cyano Analog | Dabigatran Impurity 9 | Dabigatran Impurity 8 | ethyl 3-(2-(((4-cyanophenyl)amino)methyl)-1-methyl-N-(pyridin-2-yl)-1H-benzo[d]imidazole-5-carboxami | Dabigatran Impurity E | Dabigatran Impurity 14 | Dabigatran Impurity F | Dabigatran Acid Cyano Analog | Dabigatran Impurity 9 Hydrochloride | Dabigatran Impurity 5 | Dabigatran hexilate Dimer | Dabigatran-3A-Diaryl triazinone | Dabigatran Etexilate Methylene Adduct 1 | Dabigatran Impurity 6 | Dabigatran 3-Amino4-Hydroxybenzoyl Impurity | Dabigatran Impurity 1 | Dabigatran Amide Ester Impurity | Dabigatran Impurity 3 | Dabigatran Impurity 10 | Dabigatran Impurity B | Dabigatran Etexilate O-Desethyl Impurity | Dabigatran Impurity-II oxalate | Dabigatran Impurity 22 | Dabigatran Impurity 11 | Dabigatran Impurity 74 | Dabigatran-3A-Dihexyl impurity | Dabigatran impurity J | Hexyl ((4-aminophenyl)(imino)methyl)Carbamate | Dabigatran Nitroso Impurity 17 | Dabigatran Isopropyl Ester Impurity | pentyl (Z)-(amino(4-aminophenyl)methylene)carbamate.HCl/DEM2 | DabigatranPropanoic acid impurity | Dabigatran Impurity 21 | Dabigatran Etexilate Mesylate - Impurity C (Freebase) | Dabigatran Impurity 23 | Dabigatran Impurity 19 | Des-(N-2-pyridyl-Beta-alanine Ethyl Ester) Dabigatran Etexilate 5-Ethyl Carboxylate | Dabigatran Despropionyl Impurity | Dabigatran Despropionyl Despyridinyl Impurity | Dabigatran Impurity 15 | Dabigatran Despyridinyl Impurity | Dabigatran Amido Acid Impurity-D | Dabigatran Desamino Pyridine Chloro Impurity | Dabigatran Etexilate Methylene Adduct 3 | Dabigatran Impurity 32 | Ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-1-methyl-N-(pyridine-2-yl)-1H-benzo[d]imidazole-5-ca | Dabigatran Etexilate Methylene Adduct 2 | Dabigatran Impurity 24 | ethyl 3-(3-(hydroxyamino)-4-(methylamino)-N-(pyridin-2-yl)benzamido)propanoate | diethyl 3,3'-((2,2'-((((6-oxo-1,6-dihydro-1,3,5-triazine-2,4-diyl)bis(4,1-phenylene))bis(azanediyl)) | Dabigatran Impurity 7 | Dabigatran Impurity 35 | Dabigatran Impurity 19 | Dabigatran Impurity D | Dabigatran Impurity C | Dabigatran Impurity 12 | Dabigatran impurity L | Dabigatran Impurity K | Dabigatran Impurity G | N-Nitroso Dabigatran Etexilate amide | N-(E)-Amino-Ethoxycarbonyl Dabigatran Ethyl Ester | N-Ethoxycarbonyl Dabigatran Ethyl Ester | Dabigatran Impurity 10 Hydrochloride | N-Nitroso Dabigatran | Dabigatran Ethyl Ester Impurity |