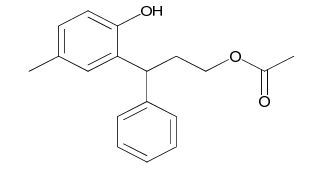
Tolterodine Diol Acetate Impurity
| Product Name | Tolterodine Diol Acetate Impurity |
|---|---|
| Alternate Names | Tolterodine Impurities, Impurities of Tolterodine |
| CAT No. | CS-O-15596 |
| CAS No. | 2514856-06-3 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 284.35 g/mol |
| Mol. For. | C₁₈H₂₀O₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Tolterodine |
| Purity | >98 |
| Smileys | OC1=C(C=C(C)C=C1)C(C2=CC=CC=C2)CCOC(C)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Tolterodine Diol Acetate Impurity is a chemical compound that is used in the pharmaceutical industry. It is a by-product that is formed during the synthesis of Tolterodine Diol Acetate, which is a medication used to treat urinary incontinence. The impurity is considered to be a minor impurity and is usually removed during the purification process of Tolterodine Diol Acetate. However, it can still be present in small amounts in the final product.
Chemically, Tolterodine Diol Acetate Impurity is a derivative of Tolterodine, which is the active ingredient in Tolterodine Diol Acetate. It is a white or off-white powder that is soluble in water and has a molecular weight of 296.39 g/mol. The structure of the impurity is similar to that of Tolterodine, but it has an additional acetate group attached to one of its hydroxyl groups.
The usage of Tolterodine Diol Acetate Impurity is limited to its role as a by-product in the synthesis of Tolterodine Diol Acetate. It has no known therapeutic or biological activity on its own. However, it is important to monitor the levels of impurities in pharmaceutical products to ensure their safety and efficacy. The presence of impurities can affect the stability and potency of the final product, and can also cause adverse effects in patients. Therefore, strict quality control measures are taken during the manufacturing process to minimize the levels of impurities in Tolterodine Diol Acetate.
Get an Instant Quote
Related Compounds
Tolterodine S-Enantiomer | Tolterodine Benzyloxy amine derivative | Tolterodine Impurity C | Tolterodine Hydrobromide Racemic | Tolterodine Acid | Tolterodine EP Impurity G (TFA Salt) | Tolterodine EP Impurity E Hydrochloride | Tolterodine Monomer | Tolterodine Lactone Impurity | rac-5-Isopropylcarbonyloxymethyl Tolterodine | Tolterodine EP Impurity E | Des Isopropyl Tolterodine Tartrate | Tolterodine Methoxy Analog Fumarate | rac-5-Hydroxymethyl Tolterodine-d5 | Tolterodine Impurity 4 | R-(+)-Tolterodine | Tolterodine Dimer | Tolterodine USP Related Compound A | Tolterodine Impurity A | Tolterodine Impurity D | Tolterodine impurity II | Tolterodine USP Related Compound A | S-(-)-Tolterodine D-Tartrate | Tolterodine EP Impurity F 2,3-dihydroxysuccinate salt | Tolterodine Monoisopropyl Analog (HCl) | Tolterodine Methoxy Propylamine Impurity Racemate | rac 5-Hydroxymethyl Desisopropyl Tolterodine | Bis-desisopropyl Tolterodine | Tolterodine Impurity G | Tolterodine Propylamine Impurity Racemate | Benzyl Tolterodine | N-Nitroso Tolterodine EP Impurity E | Benzyl R-(+)-Tolterodine |