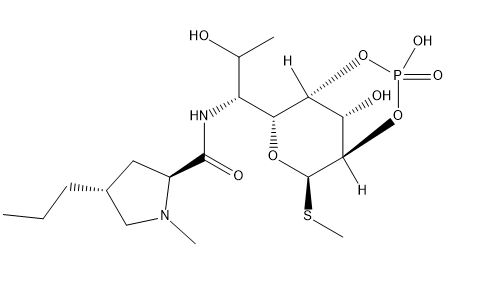
Clindamycin Impurity G
| Product Name | Clindamycin Impurity G |
|---|---|
| Alternate Names | Clindamycin Impurities, Impurities of Clindamycin |
| CAT No. | CS-O-16706 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 468.50 g/mol |
| Mol. For. | C₁₈H₃₃N₂O₈PS |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Clindamycin |
| Purity | 95% |
| Therapeutic | Antibiotics |
| Smileys | [H][C@@]12OP(O)(=O)O[C@]([H])([C@@H]1O)[C@H](O[C@@H]2SC)[C@H](NC(=O)[C@@H]1C[C@@H](CCC)CN1C)C(C)O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Clindamycin Impurity G is a degradation product of Clindamycin, which is a broad-spectrum antibiotic used to treat various bacterial infections. This impurity is formed during the manufacturing process or upon storage of the drug product. The presence of Clindamycin Impurity G in Clindamycin drug products may affect the efficacy and safety of the drug.
Chemically, Clindamycin Impurity G is an isomer of Clindamycin and has a molecular weight of 466.98 g/mol. It is known by its chemical name (2S,4R)-N-[(2R,3S,4R,5R)-5-chloro-4-hydroxy-3-methoxy-2-[(1S,2R)-2-methyl-1-[(3R,4R)-1-methyl-4-propyl-3-pyrrolidinyl]butyl]cyclohexyl]-2-hydroxy-1-methyl-3-oxobutanamide.
To ensure the safety and efficacy of Clindamycin drug products, regulatory authorities have set limits for Clindamycin Impurity G. The allowable limit for this impurity in Clindamycin drug products is not more than 0.5% of the total drug content. Therefore, it is important to monitor and control the levels of Clindamycin Impurity G during the manufacturing process and storage of Clindamycin drug products.
In conclusion, Clindamycin Impurity G is an isomer of Clindamycin that is formed during the manufacturing process or upon storage of Clindamycin drug products. It is important to monitor and control the levels of this impurity to ensure the safety and efficacy of Clindamycin drug products.
Get an Instant Quote
Related Compounds
Clindamycin 2-Phosphate Sulfoxide | Dehydro Clindamycin | Clindamycin Myristate | Clindamycin Impurity 9 | Clindamycin 4-Phosphate | Clindamycin 2-Phosphate Sulfoxide Isomer A | Clindamycin Hydrochloride EP Impurity E | Clindamycin Impurity L | Clindamycin B Palmitate | Clindamycin 4-Palmitate | Clindamycin 2,3-Dipalmitate Trifluoroacetic acid salt | Clindamycin Phosphate EP Impurity K | Isopropylidene Clindamycin Phosphate | Clindamycin 2-Phosphate Sulfone | Clindamycin Phosphate EP Impurity I | Clindamycin Sulfoxide palmitate isomer 2 | Clindamycin Sulfoxide (S-Isomer) | Clindamycin Pyrophosphate Triethylamine Salt | Clindamycin 2,4-Diphosphate | Clindamycin Palmitate Sulfoxide | Clindamycin Sulfoxide (R-Isomer) | N-Desmethyl N-Nitroso Clindamycin (mixture of isomers) | Clindamycin Phosphate Sulfone trans-N-Oxide | Clindamycin 2,3-Dipalmitate | Clindamycin Impurity (Sulfone) | Clindamycin B 3-Phosphate | Clindamycin 2-Phosphate Sulfoxide Isomer B | Clindamycin Sulfoxide palmitate isomer | Dipalmitate Impurity of Clindamycin | Clindamycin Impurity 4 | Clindamycin Phosphate N-Oxide | Clindamycin B |