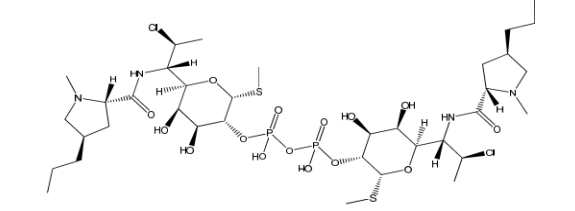
Clindamycin Phosphate EP Impurity K
| Product Name | Clindamycin Phosphate EP Impurity K |
|---|---|
| Alternate Names | Clindamycin Impurities, Impurities of Clindamycin |
| CAT No. | CS-O-16974 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 991.91 g/mol |
| Mol. For. | C₃₆H₆₆Cl₂N₄O₁₅P₂S₂ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Clindamycin |
| Purity | 95% |
| Therapeutic | Antibiotics |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Clindamycin Phosphate EP Impurity K is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for testing and quality control purposes. It is a highly pure and well-characterized compound that is used as a reference material to measure the purity and identity of Clindamycin Phosphate, an important antibiotic medication.
Clindamycin Phosphate EP Impurity K is chemically known as 7(S)-Chloro-6,7,8-trideoxy-6-[[(2S,4R)-1-methylpyrrolidin-2-yl]carbonyl]amino]-1-thio-L-threo-α-D-galacto-octopyranoside. This chemical compound is a derivative of Clindamycin Phosphate, which is a semisynthetic antibiotic that is commonly used to treat bacterial infections. The impurity K is created as a result of the manufacturing process of Clindamycin Phosphate.
In terms of usage, Clindamycin Phosphate EP Impurity K is primarily used in the development and validation of analytical methods for the detection and quantification of Clindamycin Phosphate in pharmaceutical preparations. It is also used as a reference standard in the quality control of Clindamycin Phosphate products to ensure that they meet the required standards of purity and identity.
Overall, Clindamycin Phosphate EP Impurity K is an important compound that plays a vital role in the pharmaceutical industry. Its high purity and well-characterized nature make it an essential reference material for the development, testing, and quality control of Clindamycin Phosphate products.
Get an Instant Quote
Related Compounds
N-Desmethyl N-Nitroso Clindamycin (mixture of isomers) | Clindamycin Palmitate Sulfoxide | Clindamycin 4-Palmitate | Clindamycin Impurity L | Clindamycin Sulfoxide palmitate isomer | Clindamycin Impurity 4 | Dipalmitate Impurity of Clindamycin | Clindamycin 2,4-Diphosphate | Clindamycin Pyrophosphate Triethylamine Salt | Clindamycin Phosphate N-Oxide | Clindamycin Myristate | Clindamycin 2-Phosphate Sulfoxide Isomer B | Dehydro Clindamycin | Clindamycin Sulfoxide (R-Isomer) | Clindamycin 2,3-Dipalmitate Trifluoroacetic acid salt | Clindamycin Impurity (Sulfone) | Clindamycin Impurity G | Isopropylidene Clindamycin Phosphate | Clindamycin 2-Phosphate Sulfone | Clindamycin 2-Phosphate Sulfoxide | Clindamycin 2-Phosphate Sulfoxide Isomer A | Clindamycin Sulfoxide (S-Isomer) | Clindamycin Phosphate EP Impurity I | Clindamycin Sulfoxide palmitate isomer 2 | Clindamycin Impurity 9 | Clindamycin 2,3-Dipalmitate | Clindamycin Phosphate Sulfone trans-N-Oxide | Clindamycin B 3-Phosphate | Clindamycin B | Clindamycin Hydrochloride EP Impurity E | Clindamycin B Palmitate | Clindamycin 4-Phosphate |