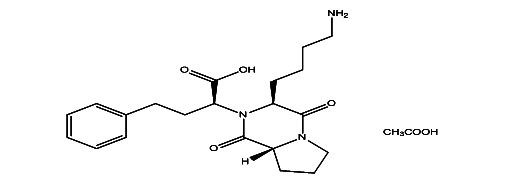
Lisinopril EP Impurity D
| Product Name | Lisinopril EP Impurity D |
|---|---|
| Alternate Names | Lisinopril Impurities, Impurities of Lisinopril |
| CAT No. | CS-O-31222 |
| CAS No. | 219677-82-4 (free base) |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 447.52 g/mol |
| Mol. For. | C₂₃H₃₃N₃O₆ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lisinopril |
| Purity | 95% |
| Therapeutic | Anti-Hypertensives |
| Smileys | O=C(O)[C@@H](N([C@H]1CCCCN)C([C@](CCC2)([H])N2C1=O)=O)CCC3=CC=CC=C3.CC(O)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lisinopril EP Impurity D is a chemical compound that is commonly used as a reference standard in analytical laboratories. It is an impurity that is typically found in small amounts in Lisinopril, which is a medication used to treat high blood pressure and heart failure. Lisinopril EP Impurity D is also known as (2S)-2-[(1S)-1-carboxy-3-phenylpropyl] hexanoic acid and is used as a reference standard for the analysis of Lisinopril.
The chemical structure of Lisinopril EP Impurity D is similar to Lisinopril, but it differs in the presence of an additional carboxylic acid group. It has a molecular weight of 344.46 g/mol and a melting point of 163-165°C. Lisinopril EP Impurity D is a white to off-white powder that is sparingly soluble in water and soluble in organic solvents such as methanol and ethanol.
In analytical laboratories, Lisinopril EP Impurity D is used in the development and validation of analytical methods for the quantification of Lisinopril and its related impurities. It is also used as a reference standard in the characterization of Lisinopril and its impurities. The use of Lisinopril EP Impurity D as a reference standard allows for accurate and precise analysis of Lisinopril, which is important for ensuring the safety and efficacy of the medication.
Get an Instant Quote
Related Compounds
Lisinopril SRS-Diastereomer | N2-(1-Ethoxycarbonyl-3-oxo-3-phenylpropyl)-N6-trifluoroacetyl-L-lysine | N-(1-Carboxy-3-phenylpropyl)-S-lisinopril (Mixture of diastereomers) | Lisinopril EP Impurity A | Lisinopril EP Impurity A | N-trifluoroacetyl Lisinopril Intermediate | Lisinopril EP Impurity E | N-Benzyloxycarbonyl Lisinopril Cyclohexyl Analogue Ethyl Methyl Diester | N-Benzyloxycarbonyl (S)-Lisinopril | N-Benzyloxycarbonyl (S)-Lisinopril Ethyl Methyl Diester | Lisinopril EP Impurity J | Lisinopril EP Impurity G | Lisinopril EP Impurity I Acetate salt | Lisinopril-D8 | Lisinopril Intermediate | Lisinopril-D4 | Lisinopril EP Impurity F | Lisinopril Des-Proline dimer - II | Lisinopril EP impurity C Acetate salt |