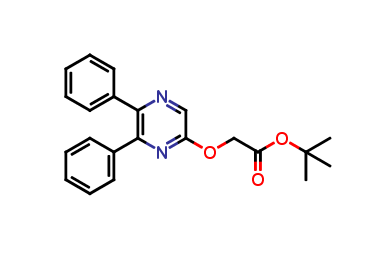
Selexipag O - Impurity
| Product Name | Selexipag O - Impurity |
|---|---|
| Alternate Names | Selexipag Impurities, Impurities of Selexipag |
| CAT No. | CS-O-32672 |
| CAS No. | 162928-03-2 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 362.42 g/mol |
| Mol. For. | C₂₂H₂₂N₂O₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Selexipag |
| Purity | 95% |
| Smileys | O=C(OC(C)(C)C)COC1=CN=C(C2=CC=CC=C2)C(C3=CC=CC=C3)=N1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Selexipag O is an impurity that is commonly found in pharmaceutical preparations of the drug Selexipag. Selexipag is a medication that is used to treat pulmonary arterial hypertension, a condition characterized by high blood pressure in the pulmonary arteries that supply blood to the lungs. Selexipag works by activating a specific receptor in the body known as the prostacyclin receptor, which helps to dilate the blood vessels and reduce blood pressure.
Selexipag O is an impurity that is formed during the synthesis of Selexipag. It is a chemical compound that is structurally similar to Selexipag but differs in its chemical composition. Selexipag O is considered an unwanted impurity in Selexipag preparations as it can alter the pharmacological properties of the drug and potentially cause adverse effects.
Chemically, Selexipag O is a derivative of Selexipag that contains a hydroxyl group (-OH) attached to the aromatic ring of the molecule. This hydroxyl group can significantly alter the physicochemical properties of the molecule and may affect its solubility, stability, and bioavailability.
In conclusion, Selexipag O is an unwanted impurity that can be found in Selexipag preparations. Its presence can affect the pharmacological properties of the drug and may cause adverse effects. Therefore, it is essential to monitor and control the levels of Selexipag O in Selexipag preparations to ensure the safety and efficacy of the medication.
Get an Instant Quote
Related Compounds
Selexipag Dimer Impurity | Selexipag Mannitol Ester | Selexipag Impurity A | Selexipag Methyl Ester Impurity | Selexipag N-Oxide | Selexipag Impurity at RT about 42.3 | Selexipag Impurity 9 | Selexipag Impurity C | Selexipag Impurity D | Selexipag Impurity B | Sexipag Glycine Adduct | Selexipag formyl impurity | Selexipag N1-Oxide | Selexipag DPB Desisopropyl Impurity | Selexipag Impurity 28 |