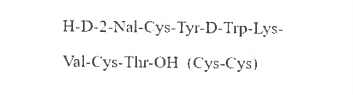Lanreotide Impurity D
| Product Name | Lanreotide Impurity D |
|---|---|
| Alternate Names | Lanreotide Impurities, Impurities of Lanreotide |
| CAT No. | CS-O-33425 |
| CAS No. | 150155-56-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 1097.31 mol/g g/mol |
| Mol. For. | C₅₄H₆₈N₁₀O₁₁S₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lanreotide |
| Therapeutic | Anti-Cancer / Oncology |
| Smileys | O=C(N[C@@](C(N[C@H]1C(C)C)=O)([H])CCCCN)[C@H](NC([C@@H](NC([C@@](CSSC[C@@H](C(N[C@H](C(O)=O)[C@H](O)C)=O)NC1=O)([H])NC([C@H](N)CC2=CC3=CC=CC=C3C=C2)=O)=O)CC4=CC=C(O)C=C4)=O)CC5=CNC6=C5C=CC=C6 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lanreotide Impurity D is a chemical compound that is used in the development and production of pharmaceutical products. It is a known impurity that is often present in Lanreotide, a medication used in the treatment of acromegaly and neuroendocrine tumors. The presence of Lanreotide Impurity D in Lanreotide can have an impact on the effectiveness and safety of the medication.
The chemical structure of Lanreotide Impurity D is similar to that of Lanreotide, but with subtle differences in the arrangement of atoms. Chemical analysis has revealed that it is a derivative of the Lanreotide molecule and is a type of deamidated peptide. This impurity has been found to be present in relatively small amounts in Lanreotide, usually less than 1%.
There is limited information available about the potential health effects of Lanreotide Impurity D, as it is not a drug that is used for medical treatments. However, it is known that the presence of impurities in medications can lead to adverse effects in patients. Therefore, pharmaceutical companies are required to monitor and control the levels of impurities in their products to ensure their safety and efficacy.
In conclusion, Lanreotide Impurity D is a chemical compound that is present in Lanreotide, a medication used in the treatment of acromegaly and neuroendocrine tumors. It is a deamidated peptide that is present in small amounts and can impact the safety and effectiveness of the medication. Pharmaceutical companies must monitor and control the levels of impurities in their products to ensure the safety of patients.
Get an Instant Quote
Related Compounds
Lanreotide Impurity I | Lanreotide Impurity X (1) | Lanreotide Impurity X (2) | Lanreotide Impurity C | Lanreotide Impurity V | Lanreotide Impurity F | Lanreotide Impurity A | Lanreotide Impurity B | Lanreotide Impurity G | Lanreotide Impurity H |