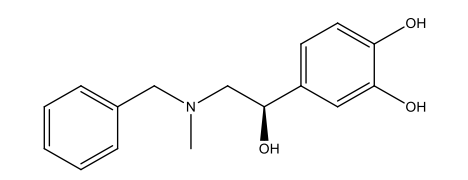
Epinephrine Impurity D
| Product Name | Epinephrine Impurity D |
|---|---|
| Alternate Names | Epinephrine Impurities, Impurities of Epinephrine |
| CAT No. | CS-O-33883 |
| CAS No. | 317351-40-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 273.33 g/mol |
| Mol. For. | C₁₆H₁₉NO₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Epinephrine |
| Smileys | O[C@H](C1=CC(O)=C(O)C=C1)CN(CC2=CC=CC=C2)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Epinephrine Impurity D, also known as 3,4-Dihydroxybenzaldehyde, is a chemical impurity that is commonly found in epinephrine formulations. This impurity is a yellowish powder with a molecular weight of 138.11 g/mol and a molecular formula of C7H6O3. It is a derivative of benzaldehyde and is formed during the synthesis of epinephrine.
Epinephrine Impurity D is not intended for use in pharmaceuticals or other healthcare products, but its presence in epinephrine formulations can have potential health implications. It is considered a potential carcinogen, and long-term exposure to this impurity may lead to adverse health effects.
To ensure the safety of epinephrine formulations, regulatory agencies such as the FDA have set limits on the amount of Epinephrine Impurity D that can be present in these products. Manufacturers of epinephrine formulations must adhere to these limits to ensure that their products are safe for use.
In conclusion, Epinephrine Impurity D is a chemical impurity that is commonly found in epinephrine formulations. While it is not intended for use in healthcare products, its presence in epinephrine formulations can have potential health implications. Regulatory agencies have set limits on the amount of this impurity that can be present in epinephrine formulations to ensure their safety.
Get an Instant Quote
Related Compounds
EPINEPHRINE IMPURITY 17 | EPINEPHRINE IMPURITY 18 | N-nitroso- norepinephrine Impurity-3 | L-Norepinephrine Hydrochloride | Norepinephrine impurity 26 hcl | Norepinephrine Impurity 40 | DL-Norepinephrine Hydrochloride | O-Methyl Norepinephrine | DL-Norepinephrine 3-Sulfate | Epinephrine Impurity 8 | N-nitroso Epinephrine | Epinephrine Impurity 12 | Epinephrine Impurity 6 | rac N-Methyl Epinephrine | N-Benzyl Epinephrine | DL-Norepinephrine 4-Sulfate | Norepinephrine Sulfonic Acid | Norepinephrine, racemic mixture | rac Epinephrine-1-Sulfuronthiate | Norepinephrine di oxo sulfonic acid impurity | a-Ethyl Norepinephrine Hydrochloride | Epinephrine Sulfate Impurity | N-nitroso- norepinephrine Impurity-2 | rac-Epinephrine-3-O-sulfate | Epinephrine 6-benzenesulfonic acid | Epinephrine Impurity 9 | Epinephrine 5- benzenesulfonic acid | Epinephrine Impuirty 2 | Deoxyepinephrine |