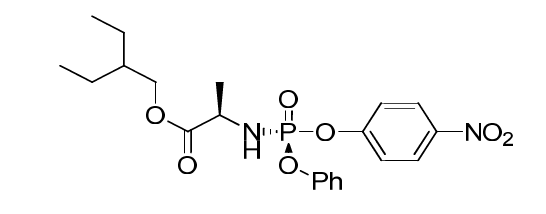
Remdesivir Impurity 3
| Product Name | Remdesivir Impurity 3 |
|---|---|
| Alternate Names | Remdesivir Impurities, Impurities of Remdesivir |
| CAT No. | CS-O-35698 |
| CAS No. | 2093124-22-0 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 450.43 g/mol |
| Mol. For. | C21H27N2O7P |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Remdesivir |
| Smileys | C[C@H](C(OCC(CC)CC)=O)N[P@@](OC1=CC=CC=C1)(OC2=CC=C([N+]([O-])=O)C=C2)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Remdesivir Impurity 3, also known as (1R,3R,4S)-3-(4-aminopyrrolo[2,1-f][1,2,4]triazin-7-yl)-2-azabicyclo[2.2.1]heptane-2-carboxylic acid, is an impurity of Remdesivir, a broad-spectrum antiviral drug that has been approved for the treatment of COVID-19. Remdesivir Impurity 3 is a byproduct that is formed during the synthesis of Remdesivir and can be isolated and purified for use as a reference standard in pharmaceutical research and development.
The chemical structure of Remdesivir Impurity 3 contains a pyrrolo[2,1-f][1,2,4]triazine moiety that is found in several antiviral agents. This moiety is believed to be responsible for the antiviral activity of Remdesivir and its derivatives. Remdesivir Impurity 3 can be used as a reference standard to study the metabolism, pharmacokinetics, and toxicity of Remdesivir in vitro and in vivo. It can also be used to develop analytical methods for the quality control of Remdesivir and its formulations.
Remdesivir Impurity 3 is a white to off-white solid that is soluble in water and organic solvents. Its purity is typically determined by high-performance liquid chromatography (HPLC) or mass spectrometry (MS) analysis. The use of Remdesivir Impurity 3 in pharmaceutical research and development is subject to strict regulatory guidelines to ensure its safety and efficacy.
Get an Instant Quote
Related Compounds
Remdesivir Impurity 2 | Remdesivir R-P isomer and S-isomer at CN | Remdesivir Impurity 1 | Remdesivir Impurity 9 | Remdesivir N-oxide | Remdesivir Phosphate Impurity | Remdesivir Bis Phosphoryl impurity | L-Alanine, N-[(S)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 4) | Remdesivir Impurity 41 | NSJ-Remdesivir-RS005 | Remdesivir Impurity 14 | Remdesivir Impurity 43 | Remdesivir Impurity 10 | Remdesivir Impurity 12 | Remdesivir Impurity 18 | L-Alanine, N-(chlorophenoxyphosphinyl)-, 2-ethylbutyl ester (IMP 7) | Remdesivir Desphenyl Impurity | L-Alanine, N-[(R)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 5) | D-Alanine, N-[(S)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 6) | GS-773095 | Remdesivir Impurity 16 | Remdesivir Desphenyl Acid Impurity | Remdesivir Impurity 8 | Remdesivir Impurity 15 | Remdesivir Impurity 11 | NSJ-Remdesivir-RS009 | Remdesivir Nucleoside Monophosphate Ammonium salt | Remdesivir impurity-18 Triethylamine salt | Remdesivir Impurity 16 | Remdesivir Impurity C | Remdesivir R-P-D Alanine Isomer | NSJ-Remdesivir-RS012 | Remdesivir Impurity 17 | NSJ-Remdesivir-RS004 | Remdesivir Impurity 13 |