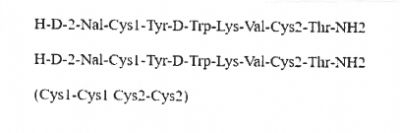Lanreotide Impurity X (1)
| Product Name | Lanreotide Impurity X (1) |
|---|---|
| Alternate Names | Lanreotide Impurities, Impurities of Lanreotide |
| CAT No. | CS-O-36396 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 2192.65 g/mol |
| Mol. For. | C₁₀₈H₁₃₈N₂₂O₂₀S₄ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lanreotide |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lanreotide Impurity X (1) is a molecular compound that is commonly used in the production of lanreotide, a medication that is used to treat acromegaly and neuroendocrine tumors. It is an impurity that is produced during the synthesis of lanreotide and is typically removed during the purification process. However, it is important to understand its chemical properties and usage in order to ensure the quality and safety of the final product.
The chemical formula of Lanreotide Impurity X (1) is C54H82N10O13S2, and its molecular weight is 1145.5 g/mol. It is a complex molecule that contains multiple functional groups, including amino acids, sulfhydryl groups, and carboxylic acids. These functional groups play an important role in the chemical properties and interactions of the compound.
In terms of usage, Lanreotide Impurity X (1) is typically considered a byproduct of the synthesis process and is not intentionally used in medical treatments. However, it is important to monitor its presence and levels during the manufacturing process to ensure that the final product is safe and effective. The impurity can potentially affect the purity and potency of the medication, so it is important to minimize its presence as much as possible.
Overall, Lanreotide Impurity X (1) is an important chemical compound that plays a critical role in the production of lanreotide. Its chemical properties and interactions must be carefully monitored in order to ensure the quality and safety of the medication.
Get an Instant Quote
Related Compounds
Lanreotide Impurity I | Lanreotide Impurity V | Lanreotide Impurity B | Lanreotide Impurity A | Lanreotide Impurity H | Lanreotide Impurity X (2) | Lanreotide Impurity F | Lanreotide Impurity C | Lanreotide Impurity D | Lanreotide Impurity G |