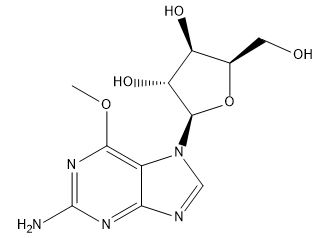
Nelarabine regio isomer impurity
| Product Name | Nelarabine regio isomer impurity |
|---|---|
| Alternate Names | Nelarabine Impurities, Impurities of Nelarabine |
| CAT No. | CS-O-37002 |
| CAS No. | 1233127-29-1 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 297.27 g/mol |
| Mol. For. | C₁₁H₁₅N₅O₅ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Nelarabine |
| Smileys | NC1=NC(OC)=C(N([C@H]2[C@@H](O)[C@H](O)[C@@H](CO)O2)C=N3)C3=N1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Nelarabine regio isomer impurity is a chemical compound that is used in the pharmaceutical industry as a reference standard for analytical testing and research purposes. This impurity is an isomer of Nelarabine, which is a medication used for the treatment of T-cell acute lymphoblastic leukemia and T-cell lymphoblastic lymphoma.
Chemically, Nelarabine regio isomer impurity is a derivative of purine nucleoside that contains a guanine base linked to an arabinose sugar. It is a white crystalline powder that is soluble in water and other polar solvents. The molecular weight of this impurity is 263.23 g/mol, and its chemical formula is C10H13N5O4.
The impurity is used to determine the quality and purity of Nelarabine drug substances and finished products. It is also used to evaluate the stability of Nelarabine formulations during the manufacturing process and storage. The impurity can be detected and quantified using various analytical techniques such as high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS).
It is important to monitor the levels of Nelarabine regio isomer impurity in Nelarabine formulations as it can affect the efficacy and safety of the medication. Therefore, the use of this impurity in the pharmaceutical industry is vital for ensuring the quality of Nelarabine drug products.
Get an Instant Quote
Related Compounds
Nelarabine α-isomer impurity | Nelarabine stage-I impurity | Nelarabine Impurity 13 | N'-(6-chloro-9-nitroso-9H-purin-2-yl)-N,N-dimethylformimidamide | Triacetyl 2-Nitro-Nelarabine | Nelarabine Desmethoxy impurity |