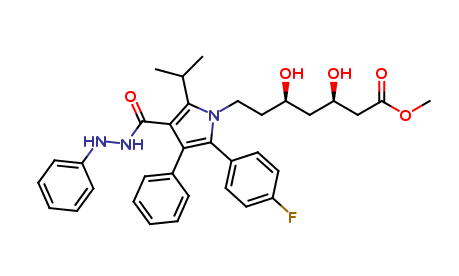
Atorvastatin diol Methyl ester impurity
| Product Name | Atorvastatin diol Methyl ester impurity |
|---|---|
| Alternate Names | Atorvastatin Impurities, Impurities of Atorvastatin |
| CAT No. | CS-O-38514 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 587.68 g/mol |
| Mol. For. | C34H38FN3O5 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Atorvastatin |
| Smileys | O=C(OC)C[C@H](O)C[C@H](O)CCN1C(C2=CC=C(F)C=C2)=C(C3=CC=CC=C3)C(C(NNC4=CC=CC=C4)=O)=C1C(C)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Get an Instant Quote
Related Compounds
sodium (3R,5R)-7-(2-(4-fluorophenyl)-5-isopropyl-3-phenyl-1H-pyrrol-1-yl)-3,5-dihydroxyheptanoate | Atorvastatin Cyclic (Fluorophenyl) Impurity | Atorvastatin Impurity D | Atorvastatin Acetonide | Atorvastatin Pyrrolidone Phenanthrene Sodium | Atorvastatin Cyclic Fluorophenyl calcium Salt Impurity | Atorvastatin Amide | Atorvastatin Impurity 1 | Atorvastatin Diepoxide Calcium Salt | Atorvastatin Impurity 19 | Atorvastatin Acid Methyl Ester | N-Nitroso Atorvastatin Impurity | Atorvastatin EP Impurity O Sodium salt | Atorvastatin EP Impurity Q sodium salt | Atorvastatin diacid impurity | Atorvastatin EP Impurity C Sodium salt | Atorvastatin 2-Hydroxy Sodium | Dihydroxy Diketo Atorvastatin Impurity | Atorvastatin Tri-acetonide tert-Butyl Ester | Atorvastatin EP Impurity A (Sodium salt) | Atorvastatin Acid t-Butyl Ester | Atorvastatin Oxirane Impurity | Atorvastatin Lactam Phenanthrene | Atorvastatin EP Impurity F (Ca Salt) | Atorvastatin Lactam Sodium Salt Impurity | Atorvastatin Pyrrolidone Analog | Atorvastatin Impurity 20 | Atorvastatin 3-Deoxy-Hept-2-Enoic Acid Ethyl Ester | Atorvastatin 3-Deoxyhept-2Z-Enoic Acid Sodium Salt | Atorvastatin EP Impurity G Sodium Salt | Atorvastatin EP Impurity F Calcium salt | Atorvastatin Dehydro Acid Sodium Salt | Allyl Ester of Atorvastatin Cyclic (Isopropyl) Impurity | Atorvastatin EP Impurity E Sodium salt | Atorvastatin Lactam Impurity calcium salt | Atorvastatin Triamino impurity | Atorvastatin EP Impurity G (3-O-methyl Atorvastatin calcium salt) | Atorvastatin Diepoxide Impurity | Atorvastatin Pyrrolidone Phenanthrene Calcium | Atorvastatin Epoxy Tetrahydrofuran Analog | Atorvastatin Diepoxide Lactone | Atorvastatin EP Impurity A | Atorvastatin EP Impurity B Calcium salt | ATV-XI | Atorvastatin Impurity F | Atorvastatin 2-Fluoro Analog | Atorvastatin Pyrrolidone Impurity | Atorvastatin Allyl Ester | Atorvastatin EP Impurity I | Atorvastatin Impurity 17 | Atorvastatin Epoxy Pyrrolooxazin tricyclic Calcium Salt | Atorvastatin Calcium Propylene Glycol Solvate | Atorvastatin Diketoene Impurity | Atorvastatin Acetonide Methyl Ester | Atorvastatin Cyclic (Fluorophenyl) Impurity | Atorvastatin EP Impurity D | Atorvastatin EP Impurity C | Atorvastatin Cyclic (Fluorophenyl) Sodium | Atorvastatin 2-Fluoro t-Butyl Ester | Atorvastatin Impurity 25 | Atorvastatin Impurity 21 | Atorvastatin 3-Deoxyhept-2E-Enoic Acid | Atorvastatin EP Impurity P (Sodium salt) | Atorvastatin Epoxy Pyrrolooxazin Analog | Atorvastatin Isopropyl Ester | Atorvastatin epoxy pyrrolooxazin 7-hydroxy analog | Atorvastatin Dehydro Sodium Salt (E/Z mixture) | Atorvastatin Ethyl Ester | Atorvastatin EP Impurity P calcium Salt | Atorvastatin Acid Sodium salt |