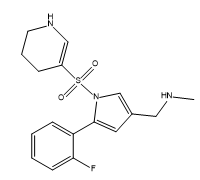
Vonoprazan Impurity 9
| Product Name | Vonoprazan Impurity 9 |
|---|---|
| Alternate Names | Vonoprazan Impurities, Impurities of Vonoprazan |
| CAT No. | CS-O-40144 |
| CAS No. | 2416241-97-7 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 349.4 g/mol |
| Mol. For. | C17H20FN3O2S |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Vonoprazan |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Vonoprazan Impurity 9 is a chemical compound that is commonly used as an impurity reference standard for vonoprazan, which is a proton pump inhibitor that is used to treat gastrointestinal disorders such as heartburn, gastroesophageal reflux disease (GERD), and gastric ulcers. Vonoprazan Impurity 9 is also known by its chemical name, (2Z)-2-[(3,5-dichloro-4-methoxyphenyl)amino]-2-oxoacetic acid.
The usage of Vonoprazan Impurity 9 is primarily for analytical purposes such as identification, quantification, and purity testing of vonoprazan in pharmaceutical formulations. This impurity is also used in research and development for the synthesis and characterization of new drugs.
The chemical information of Vonoprazan Impurity 9 includes its molecular formula, C10H8Cl2N2O4, and molecular weight, 296.09 g/mol. This compound appears as a white to off-white crystalline powder that is soluble in dimethyl sulfoxide and methanol. Its melting point is approximately 225-228°C.
In conclusion, Vonoprazan Impurity 9 is a useful tool for the pharmaceutical industry and research community for its analytical and research purposes. Its chemical properties and characteristics make it a reliable reference standard for vonoprazan and can aid in the development of new drugs for the treatment of gastrointestinal disorders.
Get an Instant Quote
Related Compounds
Vonoprazan acid impurity | N-nitroso N-des(3-pyridinylsulfonyl) Vonoprazan | Vonoprazan Impurity 4 | N-desmethyl N-ethyl vonoprazan fumarate | Vonoprazan Dimer impurity | Vonoprazan D3 | Vonoprazan pyridin-4-ylsulfonyl | Vonoprazan Nitrile impurity | N-nitroso Vonoprazan [Mixture of isomers] | Vonoprazan Impurity 6 | Vonoprazan pyridin-2-ylsulfonyl | Vonoprazan Impurity 7 | Vonoprazan Impurity 5 | Vonoprazan Impurity 45 HCl | Vonoprazan Impurity 38 | N-nitroso Vonoprazan in 1mg/mL Methanol | Vonoprazan N-Methyl Asparmic Acid Impurity | Vonoprazan Impurity 45 | Vonoprazan Impurity 43 | Vonoprazan Impurity 27 | Vonoprazan Impurity 7 HCl salt | Vonoprazan Impurity 41 | Vonoprazan Hydroxy methlyl Impuriry | Vonoprazan Impurity 2 |