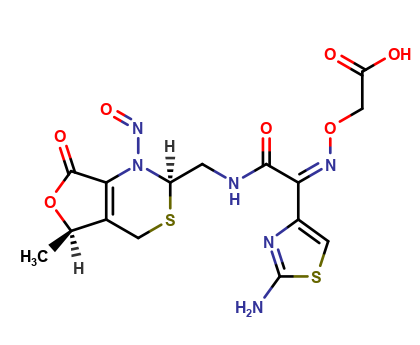
Cefixime Nitroso impurity 2
Also known as: Cefixime Nitrosamine Impurities or nitrosamine impurities of Cefixime| Product Name | Cefixime Nitroso impurity 2 |
|---|---|
| Alternate Names | Cefixime Impurities, Impurities of Cefixime |
| CAT No. | CS-O-44127 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 482.45 g/mol |
| Mol. For. | C16H14N6O8S2 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Cefixime |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Cefixime is a widely used antibiotic that belongs to the class of cephalosporins. It is used for the treatment of various bacterial infections such as pneumonia, bronchitis, urinary tract infections, and gonorrhea. However, during the manufacturing process of cefixime, impurities may be generated. One of these impurities is Cefixime Nitroso impurity 2, which is a potential carcinogen.
Cefixime Nitroso impurity 2 is a by-product of the reaction between cefixime and nitrous acid. It is a yellow to orange-colored solid that is sparingly soluble in water and organic solvents. The presence of this impurity in cefixime may reduce the efficacy of the antibiotic and may pose a risk to human health. Therefore, it is important to monitor the levels of Cefixime Nitroso impurity 2 in cefixime products and ensure they are within acceptable limits.
To detect and quantify Cefixime Nitroso impurity 2, analytical methods such as high-performance liquid chromatography (HPLC) and gas chromatography-mass spectrometry (GC-MS) are commonly used. These methods are sensitive, accurate, and reliable for the detection and quantification of impurities in pharmaceutical products.
In conclusion, Cefixime Nitroso impurity 2 is an undesirable by-product that may occur during the manufacturing process of cefixime. Its presence may affect the efficacy of the antibiotic and pose a risk to human health. Therefore, it is essential to monitor and control the levels of this impurity in cefixime products.
Get an Instant Quote
Related Compounds
Cefixime EP Impurity D | Cefixime EP Impurity C | Cefixime Impurity 1 | Cefixime related compound A | Cefixime Impurity 4 | Cefixime Acid, Thiazine Isomer Impurity, Disodium Salt | Cefixime Sulfoxide | Cefixime Impurity 4 | Cefixime D2 | Cefixime Nitroso impurity 1 | N-Acetyl Cefixime | Cefixime EP Impurity B | Cefixime tert-Butyl Ester | Cefixime glucose adduct | Cefixime related compound B | Cefixime Methyl Ester | Cefixime | Cefixime EP Impurity A (Mixture of Diastereomers) |