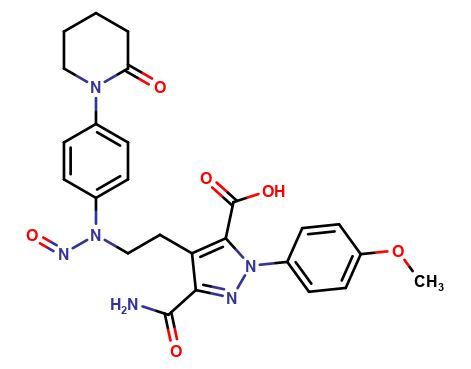
Apixaban Nitrosoacid impurity
Also known as: Apixaban Nitrosamine Impurities or nitrosamine impurities of Apixaban| Chemical Name | Apixaban Nitrosoacid impurity |
|---|---|
| Alternate Names | Apixaban Impurities, Impurities of Apixaban |
| CAT No. | CS-O-45384 |
| CAS Registry# | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 506.51 g/mol |
| Mol. For. | C25H26N6O6 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Apixaban |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Apixaban is a widely used oral anticoagulant that is used to prevent blood clots in patients with various medical conditions such as atrial fibrillation, deep vein thrombosis, and pulmonary embolism. During the manufacturing process of apixaban, impurities can be formed, one of which is the nitrosoacid impurity.
The nitrosoacid impurity is a chemical compound that is formed during the synthesis of apixaban. It is a byproduct of the reaction between apixaban and the nitrosating agent used in the manufacturing process. The nitrosoacid impurity is a potential concern as it may affect the efficacy and safety of apixaban.
The nitrosoacid impurity has been found to be present in small amounts in apixaban samples. The amount of the impurity is usually measured in parts per million (ppm). The acceptable limit for the nitrosoacid impurity in apixaban is set by regulatory authorities such as the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
To ensure the safety and quality of apixaban, manufacturers must control the formation of the nitrosoacid impurity during the synthesis process. They must also monitor the levels of the impurity in apixaban samples and ensure that they are within the acceptable limits set by regulatory authorities.
In conclusion, the nitrosoacid impurity is an important compound to consider during the manufacturing and quality control of apixaban. Manufacturers must ensure that the levels of the impurity are within acceptable limits to ensure the safety and efficacy of the drug for patients.
Get an Instant Quote
Related Compounds
Apixaban PG Ester-II | ethyl (Z)-2-chloro-2-(2-(4-methoxyphenyl)hydrazineylidene)acetate | Apixaban Impurity 28 | Apixaban Related Compound H | Hydroxy O-Demethyl Apixaban | Apixaban Related Compound 1 | Apixaban Impurity 3 | Apixaban Impurity 13 Potassium Salt | Apixaban methyl ester Dimer imp | Apixaban Impurity 26 | Apixaban Impurity 33 | Apixaban Impurity 32 | Apixaban Related Compound D | Apixaban Impurity 52 | AXN-III | Apixaban Chloro Impurity | Apixaban Ester Impurity | Apixaban Impurity 16 | Apixaban Impurity 51 | Apixaban Impurity 3 | Apixaban diacid impurity | Apixaban Impurity 12 | Apixaban Hydroxy acid Impurity | Apixaban impurity E | Apixaban impurity D | Methyl 5-((4-(3-Carbamoyl-1-(4-methoxyphenyl)-7-oxo-4,5-dihydro-1H-pyrazolo[3,4-c]pyridin-6(7H)-yl)phenyl)amino)pentanoate | Apixaban Nitrosoamide impurity | N-Nitroso 5-Chloropentanamide Apixaban | Apixaban impurity A | Apixaban Impurity - (2 H2O) | Apixaban imp Dimer-2 | Apixaban Isopropyl Ester Impurity | Apixaban Impurity 29 | Apixaban PG Ester-I | Apixaban Impurity 34 | Apixaban Amino Acid Impurity | Apixaban Methyl ester Impurity | Ethyl-2-chloro-2-(2-(4-hydroxy phenyl)hydrazono)acetate | Apixaban impurity B | Apixaban Related Compound 5 | Apixaban carboxybutylaminophenyl | Apixaban Impurity 5 | Apixaban Amino Acid Impurity Hydrochloride | O-Desmethyl Apixaban | Apixaban dimer impurity | Apixaban Impurity C | Apixaban Impurity 17 | N-(4-methoxyphenyl)nitrous hydrazide | Apixaban Impurity 44 | O-Desmethyl Apixaban Sulfate | Apixaban Impurity 24 | Desmethoxy Apixaban | Apixaban Related Compound G | Apixaban Impurity 43 | Apixaban Impurity 38 | Ethyl (2E)-2-chloro-2-[2-(4-methoxyphenyl)hydrazinylidene]acetate | Apixaban Impurity 84 | Apixaban Impurity - (3/2 H2O) | Ethyl 2-chloro-2-[2-(2-methoxyphenyl)hydrazin-1-ylidene]acetate | Apixaban Impurity 40 | Apixaban Impurity 39 | Apixaban Ethoxy Diethylamide impurity | Apixaban Impurity 27 | N-Nitroso Apixaban Amino Acid Impurity | Apixaban Impurity 14 |