Lidocaine impurity A
| Product Name | Lidocaine impurity A |
|---|---|
| Alternate Names | Lidocaine Impurities, Impurities of Lidocaine |
| CAT No. | CS-T-19784 |
| CAS No. | 87-62-7 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 121.18 g/mol |
| Mol. For. | C₈H₁₁N |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lidocaine |
| Purity | 95% |
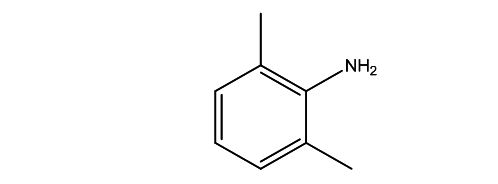
| Smileys | CC1=C(C(=CC=C1)C)N |
| Canonical Smiles | CC1=C(C(=CC=C1)C)N |
| InchIKey | UFFBMTHBGFGIHF-UHFFFAOYSA-N |
| Inchl | InChI=1S/C8H11N/c1-6-4-3-5-7(2)8(6)9/h3-5H,9H2,1-2H3 |
| IUPAC | 2,6-dimethylaniline |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lidocaine impurity A is a byproduct of the synthesis of Lidocaine, a widely used local anesthetic. It is a white or off-white crystalline powder that is sparingly soluble in water and soluble in organic solvents like methanol and ethanol. The chemical formula of Lidocaine impurity A is C14H22N2O, and its molecular weight is 234.34 g/mol.
Lidocaine impurity A is considered a minor impurity and is typically present in small amounts in Lidocaine preparations. However, its presence can affect the purity and potency of Lidocaine, and therefore, it is essential to monitor its levels during the manufacturing process.
In terms of usage, Lidocaine impurity A is not intended for direct use in any medical or pharmaceutical application. Instead, it is used as a reference standard for analytical testing and quality control purposes. Its primary use is in the development and validation of analytical methods to detect and quantify impurities in Lidocaine preparations.
In conclusion, Lidocaine impurity A is an important chemical compound that plays a significant role in ensuring the quality and safety of Lidocaine preparations. Its chemical and physical properties, as well as its usage and role in the manufacturing process, make it a crucial reference standard for analytical testing and quality control in the pharmaceutical industry.
Get an Instant Quote
Related Compounds
Ranolazine Impurity (ZEN-II) | N-Dimethyl lidocaine | Lidocaine EP Impurity J | Lidocaine EP Impurity E | Lidocaine EP Impurity I | N-Nitroso Lidocaine EP Impurity E | Lidocaine Hydrochloride Impurity H | Lidocaine Impurity E | Lidocaine EP Impurity G | Lidocaine Impurity I | Lidocaine EP Impurity D | Lidocaine EP Impurity F | Lidocaine Hydrochloride EP Impurity K | Lidocaine EP Impurity C | N-(2,6-dimethylphenyl)-2-[ethyl(methyl)amino]acetamide | Lidocaine Nitroso Impurity 1 | Methylene(bis(hydroxymethoxyphenylene))bis(phenyl) ketone |