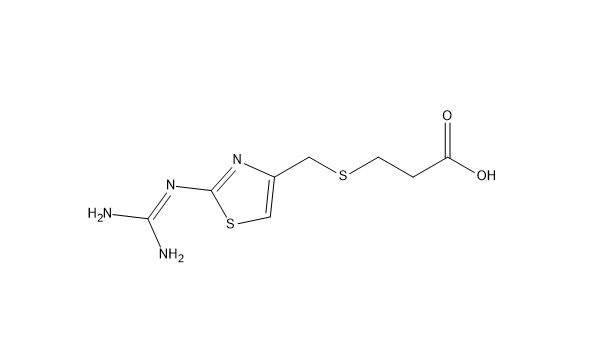
Famotidine EP Impurity F
| Product Name | Famotidine EP Impurity F |
|---|---|
| Alternate Names | Famotidine Impurities, Impurities of Famotidine |
| CAT No. | CS-T-23419 |
| CAS No. | 107880-74-0 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 260.34 g/mol |
| Mol. For. | C₈H₁₂N₄O₂S₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Famotidine |
| Therapeutic | Anti ulcer |
| Smileys | C1=C(N=C(S1)N=C(N)N)CSCCC(=O)O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Famotidine EP Impurity F is a chemical compound used in pharmaceutical research and development. Famotidine is a histamine H2 receptor antagonist used to treat gastric and duodenal ulcers, gastroesophageal reflux disease (GERD), and other conditions related to excess stomach acid production. Famotidine EP Impurity F is a known impurity of Famotidine and is often used as a reference standard for the analysis of Famotidine in pharmaceutical formulations.
Chemically, Famotidine EP Impurity F has a molecular formula of C10H11N5O2S and a molecular weight of 257.29 g/mol. It is a white to off-white powder that is soluble in methanol and slightly soluble in water. The impurity is synthesized through a series of chemical reactions involving the condensation of guanidine with various aldehydes and ketones.
In terms of usage, Famotidine EP Impurity F is used as a reference standard for the analysis of Famotidine in pharmaceutical formulations. It is also used in the development of analytical methods for the determination of Famotidine in biological samples.
Overall, Famotidine EP Impurity F plays an important role in the pharmaceutical industry by providing a reliable reference standard for the analysis of Famotidine and supporting the development of new drugs in the same therapeutic class.
Get an Instant Quote
Related Compounds
Famotidine Nitroso Impurity 1 | N-Nitroso Famotidine -II | Famotidine glucose adduct | N-sulfamoylacrylimidamide | Famotidine Adduct Impurity | Famotidine Impurity H | Famotidine Impurity 8 | N-Nitroso Famotidine - I | Famotidine EP impurity E | Famotidine EP impurity G | Famotidine Related Compound A Dihydrochloride | FAMOTIDINE IMPURITY C | Famotidine Acid Methyl Ester | Famotidine EP Impurity B (Dimaleate salt) | Dediaminosulfonyl Hydroxymethyl Famotidine | Famotidine EP impurity A hydrochloride | Famotidine Impurity 10 HCl | Famotidine EP Impurity I Hydrochloride | Famotidine Impurity 1 Hydrochloride | Famotidine Impurity I | Famotidine EP impurity D | N-Nitroso famotidine-III | Famotidine Sulfamide dimer | Famotidine EP impurity B |