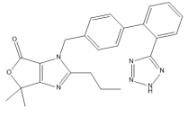
Olmesartan Medoxomil EP Impurity B
| Product Name | Olmesartan Medoxomil EP Impurity B |
|---|---|
| Alternate Names | Olmesartan Impurities, Impurities of Olmesartan |
| CAT No. | CS-T-38010 |
| CAS No. | 849206-43-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 428.49 g/mol |
| Mol. For. | C₂₄H₂₄N₆O₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Olmesartan |
| Purity | Not less than 95 % |
| Therapeutic | Anti-Hypertensives |
| Smileys | CCCC1=NC2=C(N1CC3=CC=C(C=C3)C4=CC=CC=C4C5=NNN=N5)C(=O)OC2(C)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Olmesartan Medoxomil EP Impurity B is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for analytical purposes. It is a byproduct of the synthesis of Olmesartan Medoxomil, which is an angiotensin receptor blocker that is used to treat hypertension. The chemical formula of Olmesartan Medoxomil EP Impurity B is C30H27N3O6, and its molecular weight is 525.56 g/mol.
In terms of usage, Olmesartan Medoxomil EP Impurity B is primarily used as a reference standard for the analysis of Olmesartan Medoxomil and its related impurities in pharmaceutical formulations. It is also used in research and development to study the pharmacokinetics and metabolism of Olmesartan Medoxomil in the body.
Chemically, Olmesartan Medoxomil EP Impurity B is a derivative of Olmesartan Medoxomil and belongs to the class of organic compounds known as benzimidazoles. It contains a benzimidazole ring with a carboxylic acid group and an ethyl ester group attached to it. These functional groups contribute to the compound's pharmacological properties and make it a useful analytical tool in the pharmaceutical industry.
Overall, Olmesartan Medoxomil EP Impurity B is an important chemical compound that plays a crucial role in the development and analysis of Olmesartan Medoxomil-based pharmaceuticals. Its usage and chemical properties make it a valuable tool for researchers and analysts in the field of pharmaceuticals.
Get an Instant Quote
Related Compounds
N-Nitroso Olmesartan | Olmesartan Medoxomil Impurity G | ethyl 5-(prop-1-en-2-yl)-2-propyl-1H-imidazole-4-carboxylate | Olmesartan Impurity 23 | Nitroso Olmesartan | Olmesartan Medoxomil N-Oxide | Olmesartan Methyl Ketone | Methoxy impurity of Trityl Olmesartan Medoxomil | Dehydro N2-Triphenylmethyl Olmesartan | Olmesartan medoxomil methyl ether | n-Butyl Olmesartan Medoxomil | Olmesartan Dimer Impurity | Olmesartan Nitroso Impurity 1 | Olmesartan EP Impurity C | Olmesartan Methyl Ester | Olmesartan Impurity 34 | Olmesartan Dimer Ester Impurity 1 | N-Nitroso Olmesartan Intermediate 1 | Olmesartan Medoxomil Ester Dimer | ethyl 5-acetyl-2-propyl-1-((2'-(1-trityl-1H-tetrazol-5-yl)-[1,1'-biphenyl]-4-yl)methyl)-1H-imidazole | Dehydro Olmesartan | Olmesartan Dimer Ester Impurity 2 | azide impurity of Olmesartan | N-1 Trityl Ethyl Methyl Olmesartan | Olmesartan Dimer | Olmesartan Ethyl Ester | Butyl analogue of trityl Olmesartan medoxomil | Olmesartan Medoxomil Impurity 1 | Olmesartan N1-Trityl Impurity | Olmesartan Medoxomil Impurity E | N-Trityl Olmesartan Ethyl Ester | N1-Nitrosamine Olmesartan Medoxomil | Olmesartan N2-Trityl Impurity | Olmesartan Anhydro Acid Ethyl Ester | O-Methyl Ether Olmesartan Acid | Olmesartan trityl alcohol | N-alkylated Olmesartan | Olmesartan Medoxomil EP Impurity D | Olmesartan Medoxomil EP Impurity C (1-methylethyl) | Ethyl analogue of trityl Olmesartan medoxomil | Ethyl Olmesartan Medoxomil | Dehydro Olmesartan-2-trityl Medoxomil |