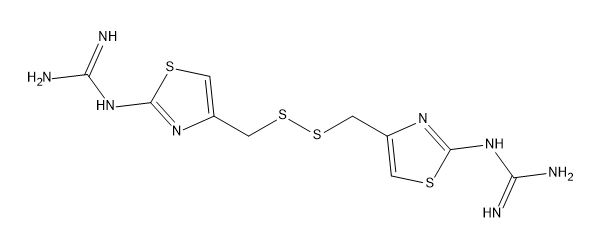
Famotidine EP impurity E
| Product Name | Famotidine EP impurity E |
|---|---|
| Alternate Names | Famotidine Impurities, Impurities of Famotidine |
| CAT No. | CS-T-49330 |
| CAS No. | 129083-44-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 374.5 g/mol |
| Mol. For. | C10H14N8S4 |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Famotidine |
| Therapeutic | Anti ulcer |
| Smileys | C1=C(N=C(S1)N=C(N)N)CSSCC2=CSC(=N2)N=C(N)N |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Famotidine EP impurity E is a chemical compound that is commonly used as a pharmaceutical intermediate in the production of Famotidine, a medication used in the treatment of ulcers and gastroesophageal reflux disease (GERD). The chemical structure of Famotidine EP impurity E is a heterocyclic compound containing a nitrogen atom and a sulfur atom, which are both important for its pharmacological activity.
Famotidine EP impurity E is typically used in small quantities during the manufacturing process of Famotidine, where it is added to the reaction mixture to facilitate the formation of the desired product. It is also used in the quality control of Famotidine, as it is often used as a reference standard to ensure the purity and potency of the final product.
From a chemical perspective, Famotidine EP impurity E is classified as a heterocyclic organic compound, which means it contains a cyclic structure that includes one or more heteroatoms (atoms other than carbon or hydrogen). The molecular formula of Famotidine EP impurity E is C7H7N3S, and its molecular weight is 165.22 g/mol.
In conclusion, Famotidine EP impurity E is an important intermediate in the manufacturing process of Famotidine, and is also used as a reference standard in quality control. Its chemical structure and properties make it a valuable component in the production of pharmaceuticals.
Get an Instant Quote
Related Compounds
Famotidine Acid Methyl Ester | Famotidine EP impurity G | Famotidine Impurity I | N-sulfamoylacrylimidamide | Famotidine EP Impurity F | Famotidine EP impurity B | Famotidine Adduct Impurity | Famotidine Nitroso Impurity 1 | Famotidine Impurity 8 | Famotidine EP impurity D | Famotidine glucose adduct | Famotidine EP impurity A hydrochloride | Dediaminosulfonyl Hydroxymethyl Famotidine | N-Nitroso Famotidine -II | N-Nitroso famotidine-III | Famotidine Related Compound A Dihydrochloride | N-Nitroso Famotidine - I | Famotidine Sulfamide dimer | Famotidine Impurity 10 HCl | Famotidine Impurity H | FAMOTIDINE IMPURITY C | Famotidine EP Impurity B (Dimaleate salt) | Famotidine Impurity 1 Hydrochloride | Famotidine EP Impurity I Hydrochloride |