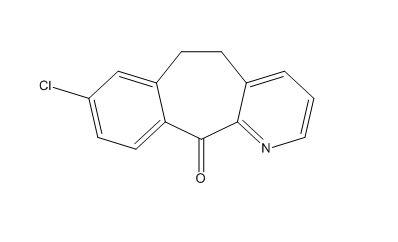
Loratadine EP Impurity B
| Product Name | Loratadine EP Impurity B |
|---|---|
| Alternate Names | Loratadine Impurities, Impurities of Loratadine |
| CAT No. | CS-T-50635 |
| CAS No. | 31251-41-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 243.69 g/mol |
| Mol. For. | C₁₄H₁₀ClNO |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Loratadine |
| Purity | Not less than 95% |
| Therapeutic | Antihistamine |
| Smileys | C1CC2=C(C=CC(=C2)Cl)C(=O)C3=C1C=CC=N3 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Loratadine EP Impurity B, also known as 7-Chloro-2-methyl-3-(4-methylpiperazin-1-yl)quinoline, is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard and impurity marker for loratadine, an antihistamine drug that is used to treat symptoms of allergies such as runny nose, watery eyes, and sneezing.
Loratadine EP Impurity B is a yellow crystalline powder that is soluble in organic solvents such as ethanol, methanol, and chloroform. It has a molecular weight of 277.8 g/mol and a melting point range of 96-98°C.
The presence of Loratadine EP Impurity B in loratadine formulations is closely monitored by pharmaceutical companies to ensure the purity and quality of the product. The impurity is typically present in small quantities, and its level is regulated by international pharmacopoeias such as the European Pharmacopoeia (EP) and the United States Pharmacopeia (USP).
In addition to its use as a reference standard and impurity marker, Loratadine EP Impurity B is also used in research and development of new drugs. Its unique chemical structure and properties make it a valuable tool in the discovery of new pharmaceutical agents for the treatment of various diseases and conditions.
Overall, Loratadine EP Impurity B is an important chemical compound in the pharmaceutical industry that plays a critical role in ensuring the purity and quality of loratadine formulations and in the development of new drugs.
Get an Instant Quote
Related Compounds
Ethyl 2-(2-oxopyrrolidin-1-yl) butanoyl carbonate | Deschloro Loratadine | Loratadine Impurity 6 | Iso Loratadine A | Loratadine EP Impurity E HCl Salt | Loratadine Impurity 15 | Loratadine LN8-bromo | Dehydro Loratadine Isomer A | Loratadine EP Impurity G | Loratadine EP Impurity E |