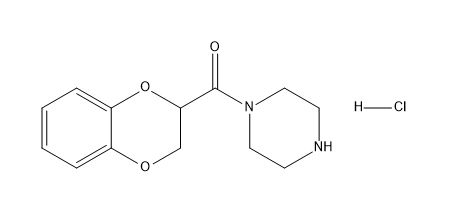
Doxazosin EP Impurity B
| Product Name | Doxazosin EP Impurity B |
|---|---|
| Alternate Names | Doxazosin Impurities, Impurities of Doxazosin |
| CAT No. | CS-T-91630 |
| CAS No. | 70918-74-0 |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 284.74 g/mol |
| Mol. For. | C₁₃H₁₇ClN₂O₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Doxazosin |
| Smileys | C1CN(CCN1)C(=O)C2COC3=CC=CC=C3O2.Cl |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Doxazosin EP Impurity B is a chemical compound that is commonly used in the pharmaceutical industry for the development of doxazosin-based medications. Doxazosin is an alpha-blocker medication that is primarily used to treat hypertension and benign prostatic hyperplasia (BPH). Doxazosin EP Impurity B is an impurity that is often produced during the synthesis of doxazosin or its derivatives.
The chemical formula of Doxazosin EP Impurity B is C23H25N3O3, and its molecular weight is 391.47 g/mol. It is a white to off-white crystalline powder that is sparingly soluble in water. The impurity has a melting point of 119-121°C and a purity of at least 98%.
The usage of Doxazosin EP Impurity B is primarily limited to the laboratory setting, where it is used as a reference standard or analytical reagent for the detection and quantification of impurities in doxazosin-based medications. Its chemical properties make it an ideal candidate for use in analytical testing, as it is stable, easily synthesized, and readily available.
While Doxazosin EP Impurity B is not intended for human consumption, it is an essential tool in the development of safe and effective doxazosin-based medications. Its chemical properties and the techniques used to synthesize and analyze it are critical in ensuring the quality and safety of doxazosin-based drugs.
Get an Instant Quote
Related Compounds
N-Descarbo(1,4-benzodioxine), N-Acetyl Doxazosin Hydrochloride | Doxazosin desethylene impurity | Doxazosin EP Impurity E | Doxazosin EP Impurity D | Doxazosin EP Impurity A | Doxazosin EP Impurity C | Doxazosin Piperazine Impurity |