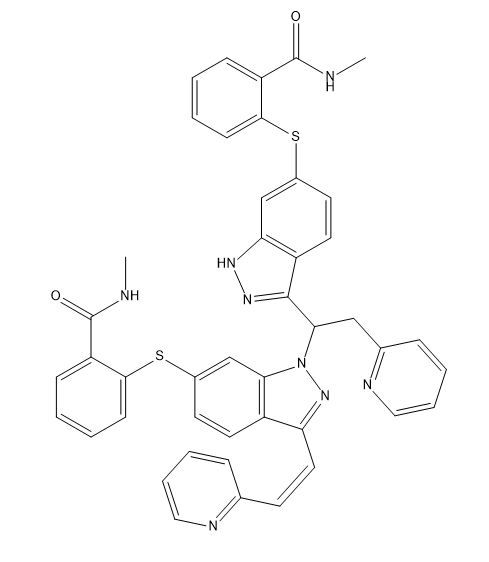
Axitinib Dimer (Asymmetric Impurity)
| Product Name | Axitinib Dimer (Asymmetric Impurity) |
|---|---|
| Alternate Names | Axitinib Impurities, Impurities of Axitinib |
| CAT No. | CS-T-93579 |
| CAS No. | 1428728-84-0 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 772.94 g/mol |
| Mol. For. | C₄₄H₃₆N₈O₂S₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Axitinib |
| Smileys | O=C(NC)C1=CC=CC=C1SC2=CC3=C(C=C2)C(/C=C\C4=NC=CC=C4)=NN3C(C5=NNC6=C5C=CC(SC7=CC=CC=C7C(NC)=O)=C6)CC8=NC=CC=C8 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Axitinib is a potent and selective inhibitor of vascular endothelial growth factor receptors (VEGFRs) that is used to treat advanced renal cell carcinoma. However, during the synthesis of axitinib, an asymmetric impurity known as axitinib dimer can form. This impurity can negatively affect the purity and efficacy of the final product, and must be carefully monitored and controlled during the manufacturing process.
Axitinib dimer is a complex organic molecule with a molecular weight of 788.9 g/mol. Its chemical formula is C42H41N7O5, and its structure consists of two axitinib molecules that are linked together through a nitrogen atom. This results in a molecule that is larger and more complex than axitinib itself, with different physicochemical properties and potential biological effects.
Due to its potential impact on the quality and safety of axitinib, axitinib dimer is closely monitored and controlled during the manufacturing process. Analytical methods such as high-performance liquid chromatography (HPLC) and mass spectrometry are used to detect and quantify the impurity, and various process parameters are adjusted to minimize its formation.
Overall, axitinib dimer is an important impurity that must be carefully managed during the production of axitinib. By ensuring that the final product is of high purity and efficacy, patients can receive the maximum benefit from this important cancer drug.
Get an Instant Quote
Related Compounds
Axitinib Impurity 6 | Axitinib Des Iodo analog | Axitinib N-Oxide | Axitinib Impurity 8 | Axitinib Sulfonyl Impurity | Axitinib Impurity 9 | Axitinib Impurity 8 | Axitinib Impurity 27 | Axitinib adduct -1 | Dehydro Axitinib | Axitinib EP Impurity D | Axitinib Dimer | Axitinib Z-isomer | Axitinib N-Formyl impurity | Axitinib Impurity 2 | N-Nitroso Axitinib | Axitinib Impurity 4 |