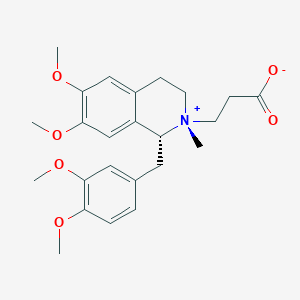
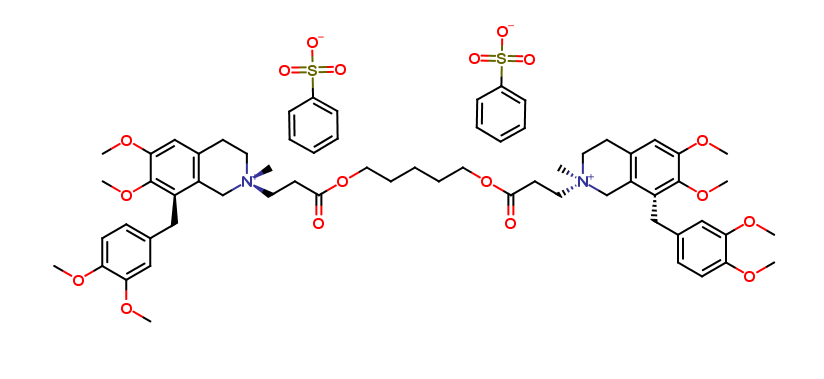
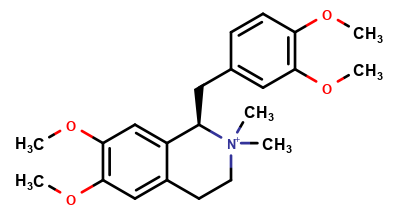
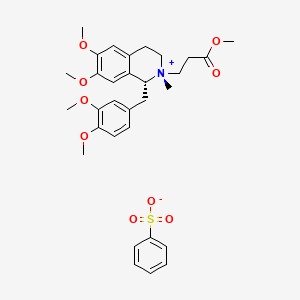
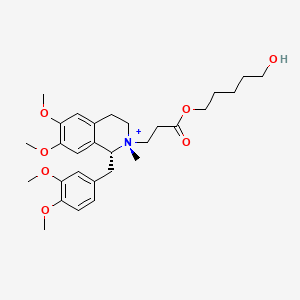
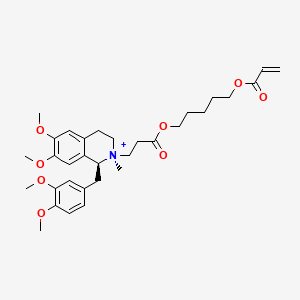
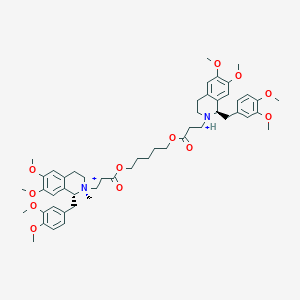
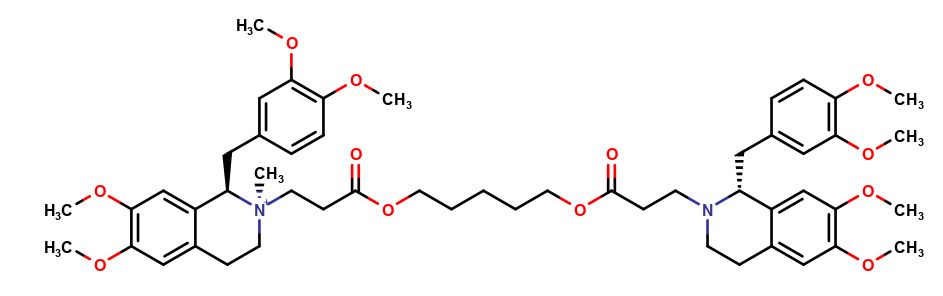
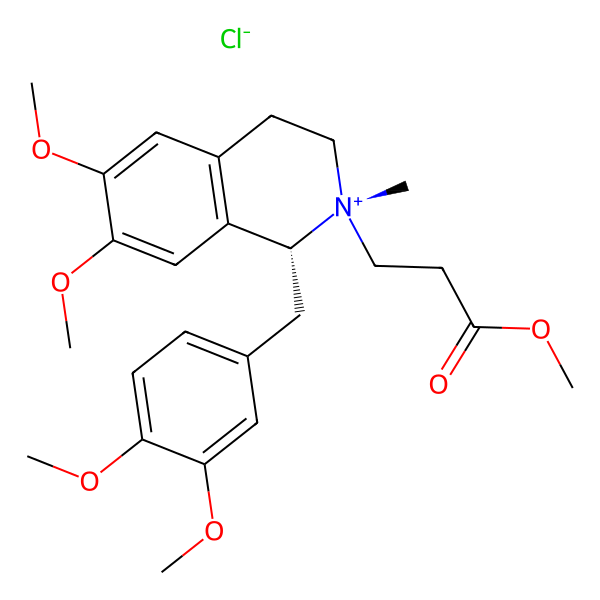
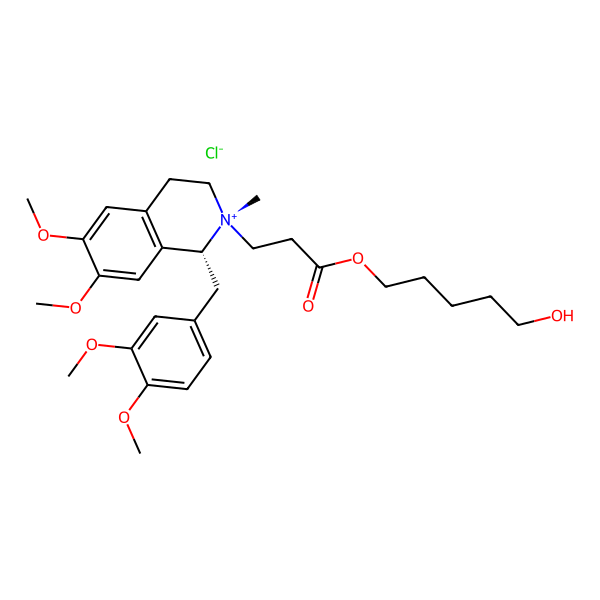
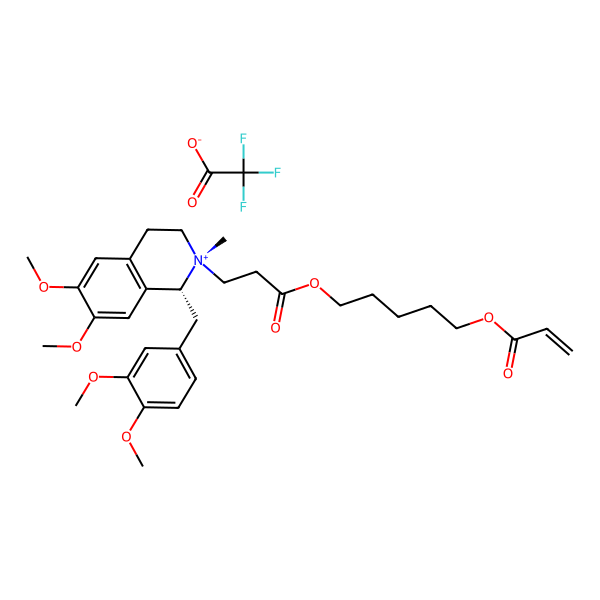
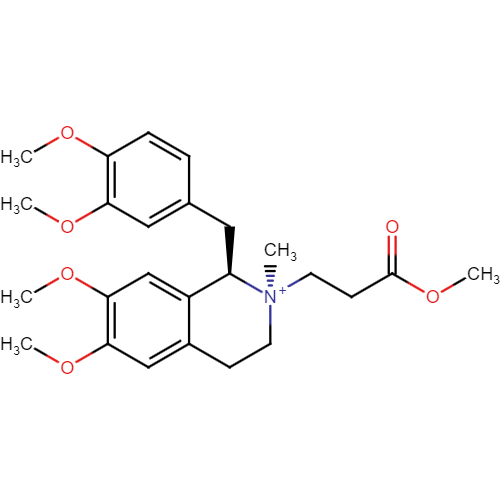
Cisatracurium Impurities and its Related Products
Cisatracurium Impurities refer to any substances or compounds that are present in a sample of cisatracurium other than the active pharmaceutical ingredient (API). These impurities may arise during the manufacturing process or due to other factors such as storage conditions. It is important to identify and quantify these impurities to ensure the safety and efficacy of the drug. Regulatory agencies such as the FDA require strict limits on the levels of impurities in drug products.