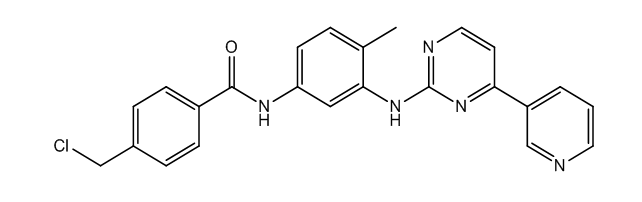
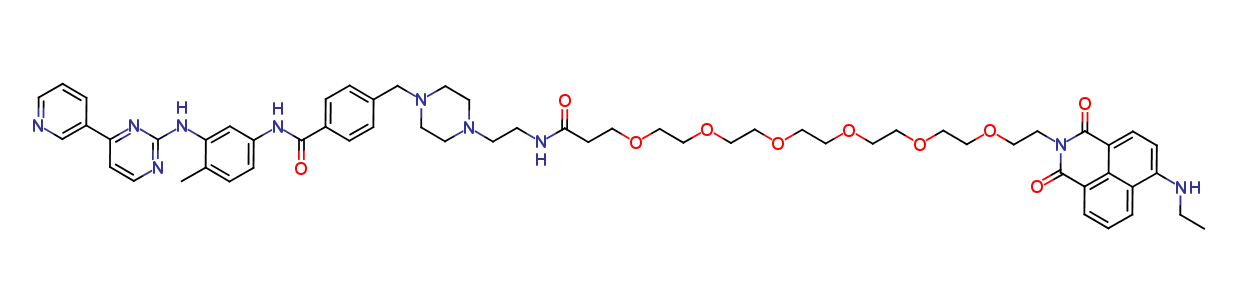
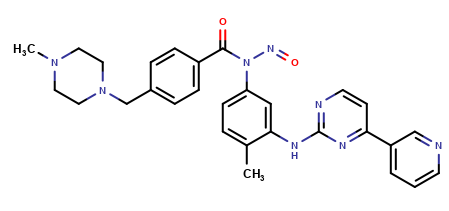
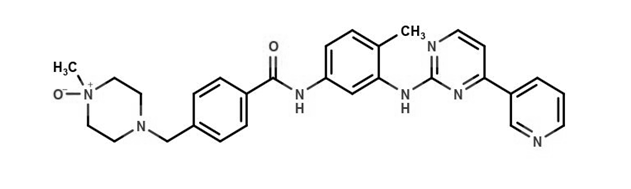
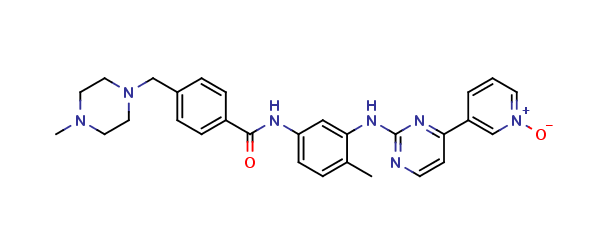
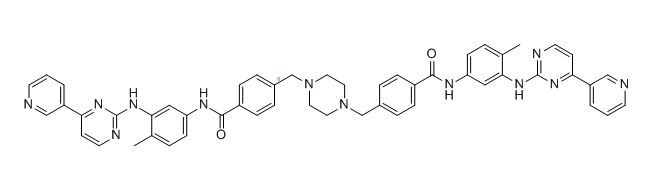
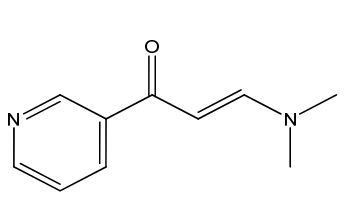
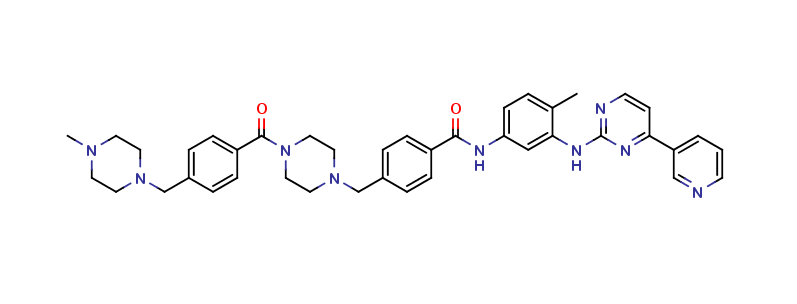
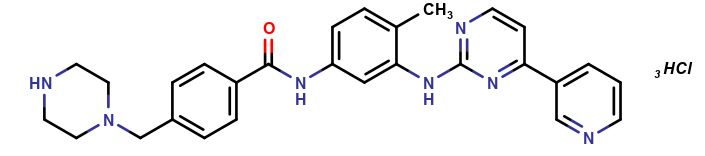
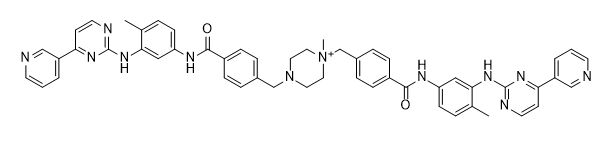
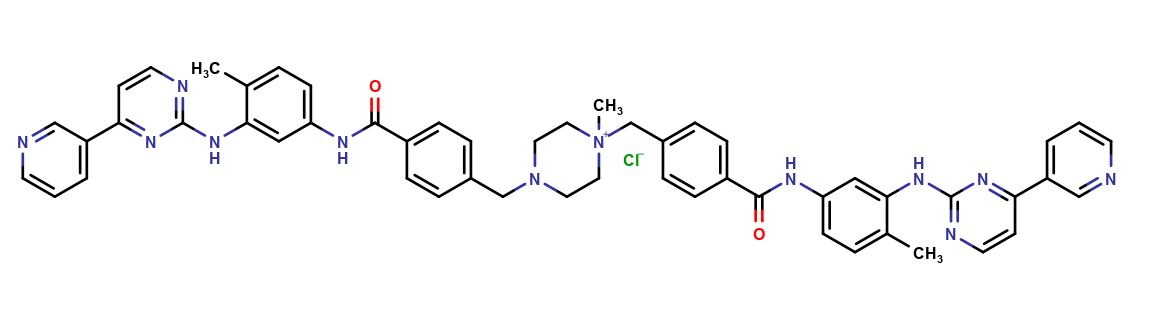
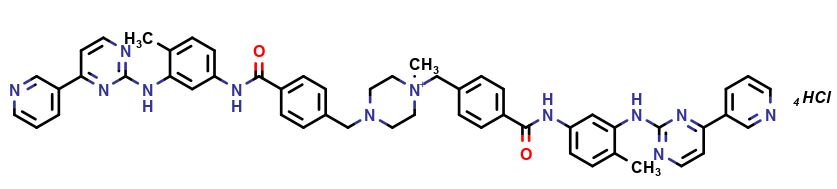
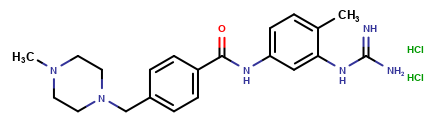
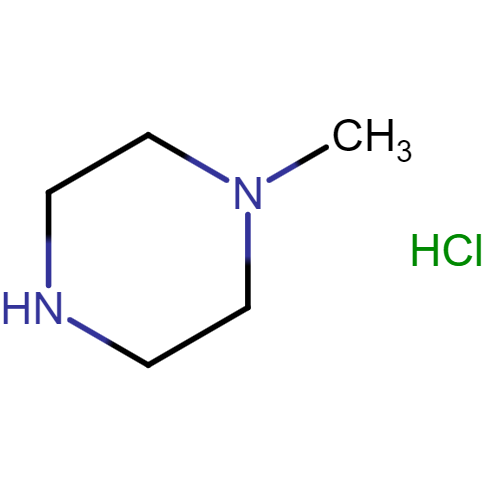
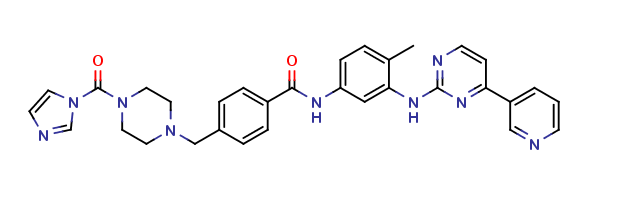
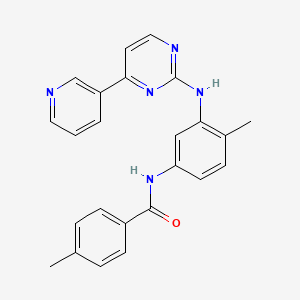
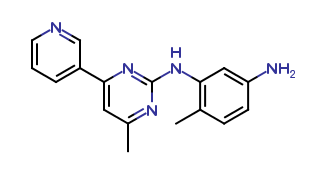
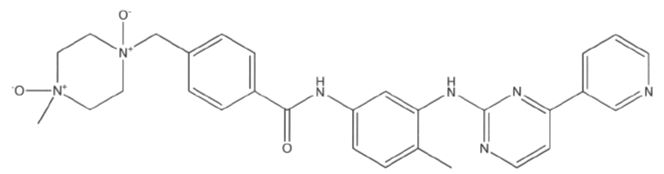
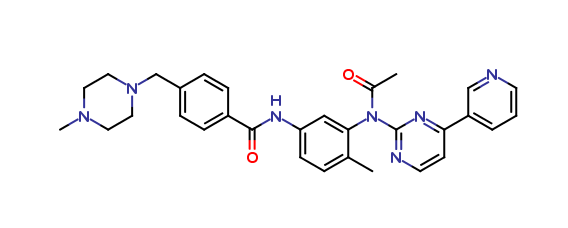
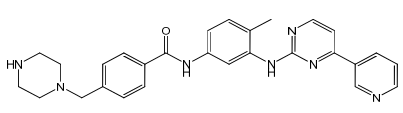
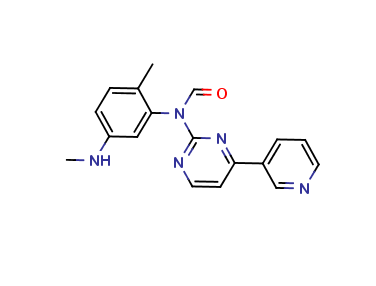
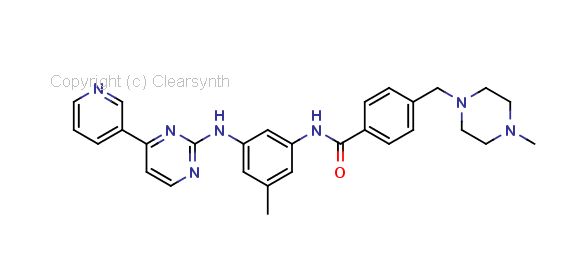
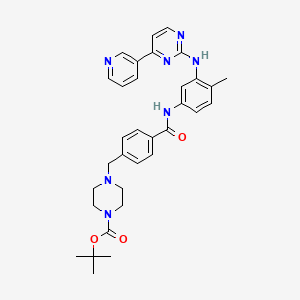
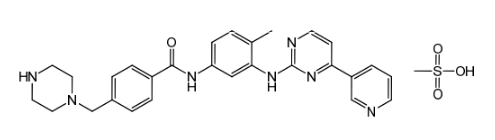
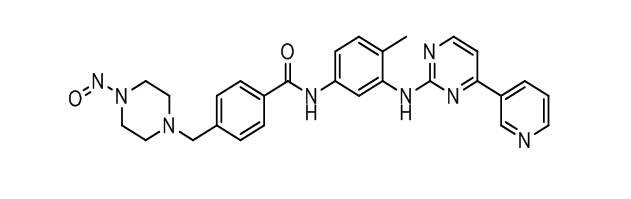
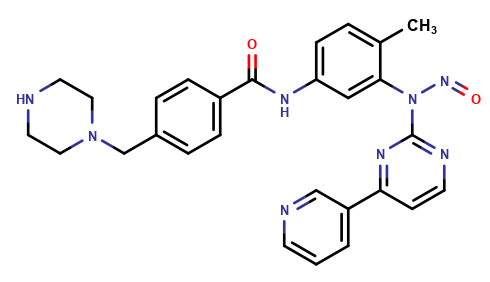
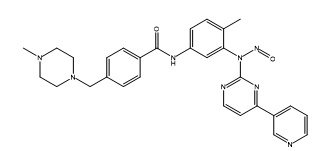
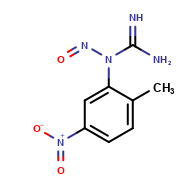
Imatinib Impurities and its Related Products
Imatinib Impurities are the unwanted substances or by-products that can potentially be present in Imatinib drug formulations. These impurities can arise during the synthesis, manufacturing, or storage of the drug and may pose a risk to patient safety. Therefore, it is essential to detect, identify, and control these impurities in order to ensure the quality, safety, and efficacy of Imatinib drug products. Various analytical techniques, including HPLC, GC, LC-MS, and NMR, are used to detect and quantify these impurities in Imatinib formulations.


![Imatinib Impurities 4-[(4-Methylpiperazin-1-yl)methyl]benzoic acid dihydrochloride](https://clearsynth.com/structure/4---4-Methylpiperazin-1-yl-methyl-benzoic-acid-dihydrochloride.png)