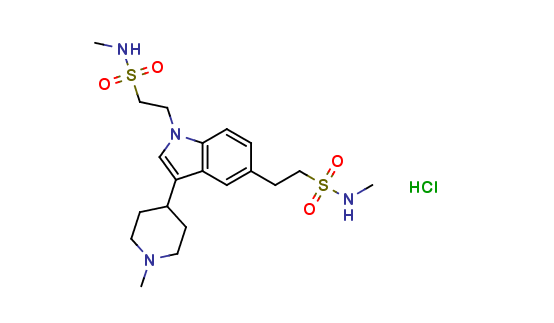
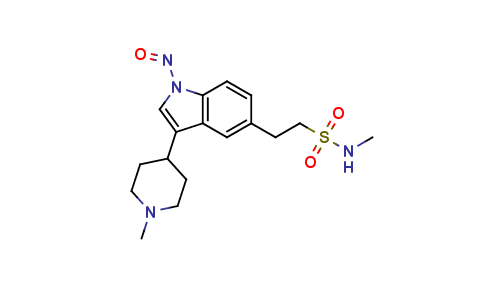
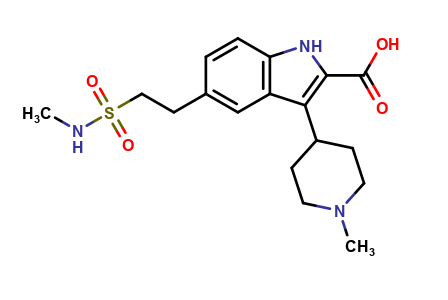
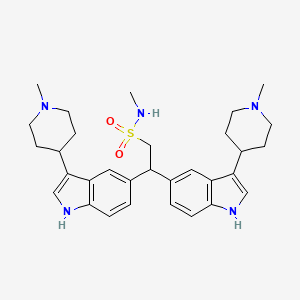
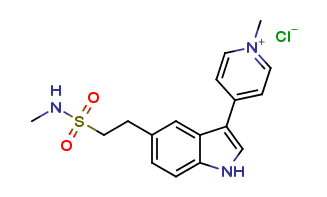
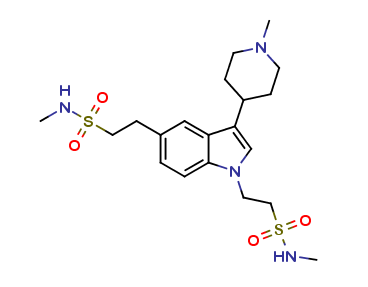
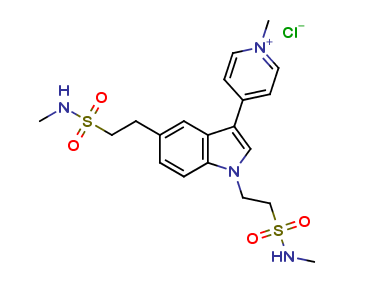
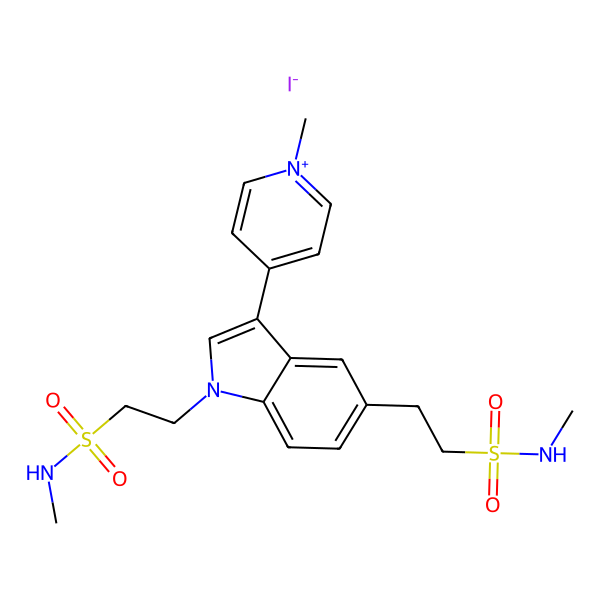
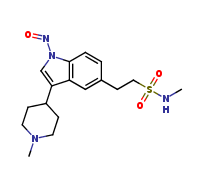
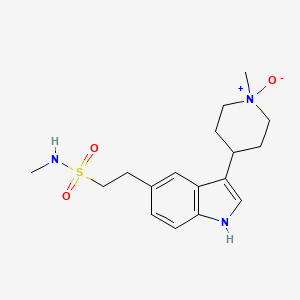
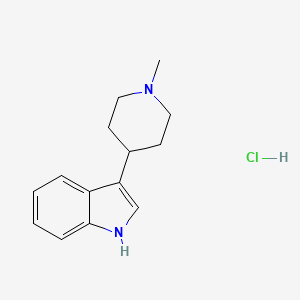
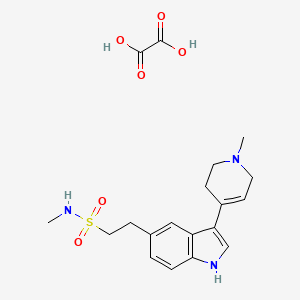
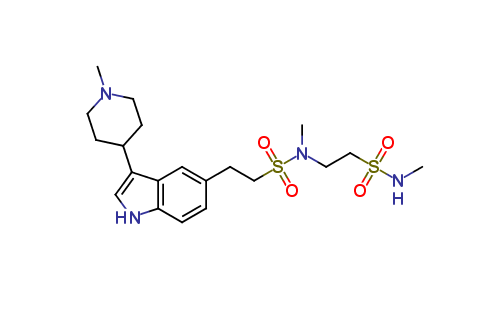
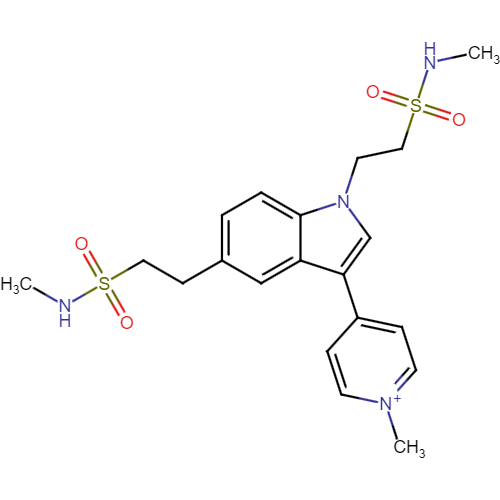
Naratriptan Impurities and its Related Products
Naratriptan Impurities refer to any undesired substances that may be present in Naratriptan drug substances or products. These impurities can arise from various sources such as synthesis, degradation, or storage conditions. Proper characterization and quantification of these impurities are essential to ensure the safety and efficacy of Naratriptan medications. Regulatory authorities have established guidelines for the acceptable limits of impurities in pharmaceuticals, and manufacturers must comply with these requirements to ensure patient safety.