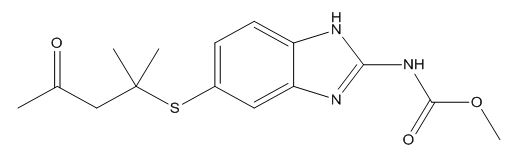
Albendazole EP Impurity H
| Product Name | Albendazole EP Impurity H |
|---|---|
| Alternate Names | Albendazole Impurities, Impurities of Albendazole |
| CAT No. | CS-O-32474 |
| CAS No. | 2469260-71-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 321.39 g/mol |
| Mol. For. | C₁₅H₁₉N₃O₃S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Albendazole |
| Purity | >98 |
| Therapeutic | Anti-Fungals |
| Smileys | O=C(OC)NC1=NC2=CC(SC(CC(C)=O)(C)C)=CC=C2N1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Albendazole EP Impurity H is a chemical compound that is commonly used as a reference standard in various pharmaceutical and research applications. It is a highly pure and stable substance that is used to identify and quantify impurities in albendazole drug formulations.
Chemically, Albendazole EP Impurity H is also known as methyl (5-(propylthio)-1H-benzimidazol-2-yl)carbamate. It is a white to off-white crystalline powder that is sparingly soluble in water, but soluble in organic solvents such as ethanol, methanol, and acetonitrile.
The primary use of Albendazole EP Impurity H is in quality control and analytical testing of albendazole drug products. It is used as a reference standard to ensure that the drug product meets the required specifications for purity and efficacy. It is also used in research for the development of new drugs and formulations.
In terms of safety, Albendazole EP Impurity H is considered to be a low-risk chemical. It is not classified as a hazardous substance and does not pose any significant health or environmental risks when handled and used according to standard safety protocols.
Overall, Albendazole EP Impurity H is a valuable tool in the pharmaceutical and research industries, helping to ensure the safety and effectiveness of albendazole drug products and contributing to the development of new and improved treatments.
Get an Instant Quote
Related Compounds
Albendazole EP Impurity A | Albendazole EP Impurity F | Albendazole EP Impurity L | Albendazole EP Impurity J | ALBENDAZOLE POLYMORPH FORM II | Calcium Cyanamide | Albendazole Impurity 7 (Methyl Cyanocarbamate) | Albendazole Impurity 10 | Albendazole EP Impurity K | Albendazole Impurity 2 | N-Nitroso Albendazole | Albendazole EP Impurity B | Albendazole EP Impurity I | Albendazole Impurity 1 | Albendazole EP Impurity E | Albendazole EP Impurity D | ALBENDAZOLE POLYMORPHFORM I | Albendazole EP Impurity I Sulfate Salt | Albendazole EP Impurity G | Albendazole Impurity 3 (2-Nitro-4-(Propylthio)-Aniline) | Ethyl Albendazole | Albendazole sulfoxide | Albendazole-2-aminosulfone Hydrochloride |