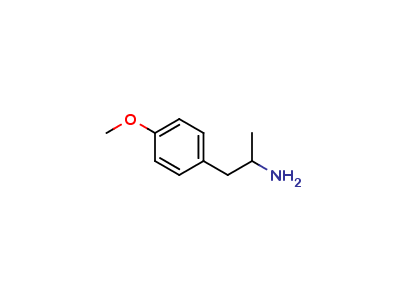
Formoterol Impurity G
| Product Name | Formoterol Impurity G |
|---|---|
| Alternate Names | Formoterol Impurities, Impurities of Formoterol |
| CAT No. | CS-O-31109 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 165.23 g/mol |
| Mol. For. | C₁₀H₁₅NO |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Formoterol |
| Purity | Not less than 90 % |
| Therapeutic | Anti-Asthma / COPD |
| Smileys | CC(N)CC1=CC=C(OC)C=C1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Formoterol Impurity G is a synthetic organic compound that belongs to the class of pharmaceutical impurities. This impurity is used in the pharmaceutical industry as a reference standard for the identification, quantification, and characterization of Formoterol, which is a long-acting beta-agonist bronchodilator used for the treatment of asthma and chronic obstructive pulmonary disease (COPD). Formoterol Impurity G is also used for the development and validation of analytical methods for the analysis of Formoterol and its related substances.
The chemical name of Formoterol Impurity G is (R)-6-(4-hydroxyphenyl)-2,5,7-trioxo-2,5,6,7-tetrahydro-1H-pyrrolo[3,4-c]pyridine-3-carboxylic acid methyl ester. Its molecular formula is C16H12N2O6 and its molecular weight is 328.27 g/mol. The compound is a white to off-white powder that is soluble in water, methanol, and acetonitrile.
Formoterol Impurity G is a degradation product of Formoterol that forms during the manufacturing process or storage of the drug product. It is important to monitor the levels of this impurity in Formoterol drug substances and drug products to ensure their quality, safety, and efficacy. The International Conference on Harmonization (ICH) guidelines recommend that the level of Formoterol Impurity G should not exceed 0.5% in Formoterol drug substances and drug products.
Get an Instant Quote
Related Compounds
Formoterol Imine Impurity | Formoterol Impurity 13 | Formoterol Impurity 1 | Formoterol Fumarate EP Impurity C | Formoterol Fumarate Dihydrate EP Impurity I (R,S-isomer) ((R,S)-Formoterol) | Formoterol EP Impurity C | Formoterol impurity F fumarate salt | Formoterol Fumarate EP Impurity E | Formoterol Fumarate EP Impurity I | Formoterol Fumarate EP Impurity D | Formoterol impurity B fumarate salt | Formoterol impurity D fumarate salt | Formoterol impurity E fumarate salt | rac-O-Benzyl N-Phenyl Formoterol(Mixture of Diastereomers) | Formoterol impurity C fumarate salt | N-(5-((R)-2-(benzyl((R)-1-(4-methoxyphenyl)propan-2-yl)amino)-1-hydroxyethyl)-2-hydroxyphenyl)formamide | Formoterol Fumarate dihydrate Impurity | Arformoterol Unknown Impurity at 0.97RRT Impurity | Formoterol Impurity D | Formoterol Impurity 2 | Formoterol Related Compound C | Formoterol EP Impurity A Fumarate | N-nitroso Formoterol | Formoterol Ethyl Ether Impurity | Formoterol Fumarate EP Impurity B | Formoterol related compound I | C-Demethyl N-Benzyl Formoterol O-Benzyl Ether | Arformoterol Impurity 1 | Formoterol Fumarate Dihydrate EP Impurity I (S,R-isomer) ((S,R)-Formoterol) | Formoterol Fumarate EP Impurity A | Formoterol Fumarate EP Impurity F | Formoterol Impurity G Hydrochloride | Formoterol Fumarate EP Impurity H | Formoterol Fumarate Dihydrate - Impurity H (Freebase) |