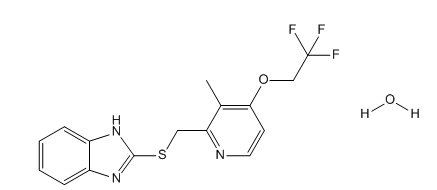
Lansoprazole Related Compound B
| Product Name | Lansoprazole Related Compound B |
|---|---|
| Alternate Names | Lansoprazole Impurities, Impurities of Lansoprazole |
| CAT No. | CS-O-37898 |
| CAS No. | 1288338-69-1 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 371.38 g/mol |
| Mol. For. | C₁₆H₁₆F₃N₃O₂S |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lansoprazole |
| Smileys | FC(F)(F)COC1=C(C)C(CSC2=NC3=CC=CC=C3N2)=NC=C1.[H]O[H] |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lansoprazole Related Compound B (LRB) is a chemical compound that is closely related to Lansoprazole, a medication that is used to treat gastric acid-related disorders such as gastroesophageal reflux disease (GERD), peptic ulcer disease, and Zollinger-Ellison syndrome. LRB is an impurity that is found in small amounts in Lansoprazole formulations, and it is important to monitor its levels to ensure the quality and efficacy of the medication.
LRB is a white to off-white crystalline powder that is soluble in organic solvents such as methanol, ethanol, and acetonitrile. It has a molecular weight of 369.41 g/mol and a chemical formula of C18H14F3N3O2S. LRB is a proton pump inhibitor (PPI) that works by reducing the production of acid in the stomach, thus relieving the symptoms of acid-related disorders.
LRB is typically analyzed using high-performance liquid chromatography (HPLC) methods, which can detect and quantify its levels in Lansoprazole formulations. The acceptable limit for LRB in these formulations is typically less than 0.1%, which ensures that the medication is safe and effective for use.
In conclusion, LRB is an important impurity that is closely related to Lansoprazole, and its levels must be monitored to ensure the quality and efficacy of the medication. HPLC methods are typically used to analyze LRB, and the acceptable limit for its presence in Lansoprazole formulations is less than 0.1%.
Get an Instant Quote
Related Compounds
Lansoprazole Impurity 33 | Lansoprazole Related Compound 4 | N-[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl]methyl Lansoprazole Sulfone | Lansoprazole D3 | Destrifluoro ethoxy Lansoprazole | Nitroso Lansoprazole | Lansoprazole Related Compound 5 | Lansoprazole Sulfide Impurity | Lansoprazole Pyridinone impurity | Lansoprazole Desulphur Impurity | Lansoprazole Impurity 1 | Lansoprazole Thioxo Impurity | N-[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl]methyl Lansoprazole | Lansoprazole impurity(M+467) | N-Nitroso Lansoprazole sulfide | Lansoprazole Sulfide N-Oxide | Lansoprazole Related Compound 6 | Lansoprazole Cyclized Impurity | Lansoprazole Base hydrolysis | N-[3-Methyl-4-(2,2,2-trifluoroethoxy)-2-pyridinyl]methyl Lansoprazole Sulfide |