Ofloxacin EP impurity E
| Product Name | Ofloxacin EP impurity E |
|---|---|
| Alternate Names | Ofloxacin Impurities, Impurities of Ofloxacin |
| CAT No. | CS-O-11038 |
| CAS No. | 82419-52-1 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 347.34 g/mol |
| Mol. For. | C₁₇H₁₈FN₃O₄ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ofloxacin |
| Purity | 95% |
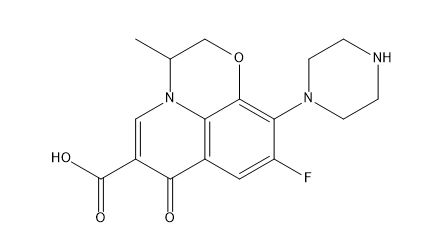
| Smileys | CC1COC2=C3N1C=C(C(=O)C3=CC(=C2N4CCNCC4)F)C(=O)O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ofloxacin EP impurity E is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for the analysis and identification of Ofloxacin. Ofloxacin is a fluoroquinolone antibiotic that is used to treat a variety of bacterial infections in humans, such as respiratory tract infections, urinary tract infections, and skin infections. Ofloxacin EP impurity E is a known impurity that can be present in Ofloxacin formulations, and its presence can affect the quality and efficacy of the drug.
The chemical name of Ofloxacin EP impurity E is 7-[(1R,2S)-2-fluorocyclopropyl]-4-oxo-1,4-dihydroquinoline-3-carboxylic acid. Its molecular formula is C14H11FNO3, and its molecular weight is 265.24 g/mol. Ofloxacin EP impurity E is a white to off-white crystalline powder that is soluble in dimethyl sulfoxide, sparingly soluble in methanol, and practically insoluble in water.
The usage of Ofloxacin EP impurity E is primarily for analytical purposes in the pharmaceutical industry. It is used as a reference standard in the development and validation of analytical methods for the detection and quantification of Ofloxacin and its related impurities. The information obtained from these analytical methods is used to ensure the quality, safety, and efficacy of Ofloxacin formulations.
Get an Instant Quote
Related Compounds
Ofloxacin Dihydrooxazole Impurity | Ciprofloxacin Impurity I HCl | Desmethyl Dextrofloxacin | Ofloxacin Impurity A | N-Nitroso N-Desmethyl Ofloxacin | Ciprofloxacin Impurity 11 | Ciprofloxacin N-Oxide | Ciprofloxacin Impurity 9 | Ciprofloxacin EP Impurity E | Ciprofloxacin Impurity 4 | Ofloxacin EP Impurity D Ethyl Ester | Ciprofloxacin Impurity 8 | N-Desmethyl Ofloxacin | Ofloxacin Desmethylpiperazin Methylester | Ofloxacin N-oxide | N-Desmethyl Danofloxacin | Ciprofloxacin Impurity 10 | Ciprofloxacin EP Impurity E HCl | N-Nitroso Ciprofloxacin | Ciprofloxacin EP Impurity B | Ciprofloxacin Methoxy Analog | N-Ethoxycarbonyl Ciprofloxacin | Ciprofloxacin Impurity 5 | Desmethyl Ofloxacin Hydrochloride | N,N’-Desethylene Ofloxacin Hydrochloride | N-Formyl Ciprofloxacin | Ciprofloxacin EP Impurity F | N-Nitroso Ofloxacin tetrafluorobenzoyl intermediate | Ciprofloxacin Impurity 7 | Ciprofloxacin EP Impurity D |