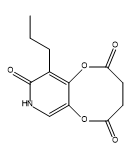
Oseltamivir Nitroso Impurity 1
Also known as: Oseltamivir Nitrosamine Impurities or nitrosamine impurities of Oseltamivir| Product Name | Oseltamivir Nitroso Impurity 1 |
|---|---|
| Alternate Names | Oseltamivir Impurities, Impurities of Oseltamivir |
| CAT No. | CS-EO-02085 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 356.46 g/mol |
| Mol. For. | C₁₈H₃₂N₂O₅ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Oseltamivir |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Oseltamivir Nitroso Impurity 1 is a chemical compound that is commonly used in the pharmaceutical industry. It is used as an impurity in the production of Oseltamivir, a drug that is used to treat influenza viruses. Oseltamivir Nitroso Impurity 1 is formed during the synthesis of Oseltamivir and hence it is important to have a good understanding of its chemical properties and usage.
Oseltamivir Nitroso Impurity 1 is a nitroso compound that has a molecular weight of 235.2 g/mol. It is a yellowish powder that is soluble in water and ethanol. The compound is highly reactive and has a short half-life, which makes it difficult to isolate and study. Despite this, it is an important impurity in the production of Oseltamivir, as it can affect the efficacy and safety of the final product.
The presence of Oseltamivir Nitroso Impurity 1 in the final product can lead to adverse effects on patients, including liver toxicity and allergic reactions. Therefore, it is important to monitor and control the levels of this impurity during the production of Oseltamivir. Various techniques such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS) are used for the detection and quantification of Oseltamivir Nitroso Impurity 1.
In conclusion, Oseltamivir Nitroso Impurity 1 is a significant impurity in the production of Oseltamivir. It is important to monitor and control its levels during the manufacturing process to ensure the safety and efficacy of the final product.
Get an Instant Quote
Related Compounds
Oseltamivir Impurity 31 | Oseltamivir Impurity 6 | Oseltamivir EP Imp U | ent-Oseltamivir Phosphate | Oseltamivir Impurity 15 | OSELTAMIVIR CITRIC ACID ADDUCT 2 | Oseltamivir EP Imp R | Oseltamivir Impurity 52 | Oseltamivir Related Compound B | Oseltamivir Phosphate Impurity B | Oseltamivir glucose adduct-2 | Oseltamivir Acid Methyl Ester | Oseltamivir-Glucose Adduct 1 | OSELTAMIVIR CITRIC ACID ADDUCT II | Oseltamivir Impurity 15 | Oseltamivir EP Impurity B | Oseltamivir Related Compound A | Oseltamivir Impurity 32 | Oseltamivir Impurity 28 | Oseltamivir Impurity 14 | Oseltamivir Impurity 4 | Oseltamivir 5-Desamino 5-(2-Formyl 5-Hydroxymethylpyrrol-1-yl) | Oseltamivir impurity A | Oseltamivir Impurity 19 | Oseltamivir RRR Isomer | Oseltamivir RSR Isomer | Oseltamivir Pyrrole analog | Oseltamivir Impurity 25 | Acetamido azidesulfonamide | Oseltamivir Impurity 48 | Oseltamivir Impurity 8 | OSELTAMIVIR CITRIC ACID ADDUCT | Oseltamivir Acid Methyl Ester Phosphate Salt | Ethyl (3S, 4S, 5S)-4-acetamido-5-amino-3-(1- ethyl propoxy) cyclohex-1-ene-1-carboxylate | Oseltamivir Impurity 26 | Oseltamivir Impurity 7 | Oseltamivir RSS Isomer | Oseltamivir Nitroso Impurity 2 | Oseltamivir Impurity 24 | Rac- Oseltamivir hydroxymethyl furan | Oseltamivir diallyl amine impurity | Oseltamivir EP Impurity E HCl | Oseltamivir Impurity 23 | ethyl 4-(pentan-3-yloxy)benzoate | N-Nitroso Oseltamivir impurity | Oseltamivir Impurity 41 | Oseltamivir Impurity 27 | Oseltamivir Impurity 22 | Rel 4-N-Desacetyl-5-N-acetyl Oseltamivir Acid |