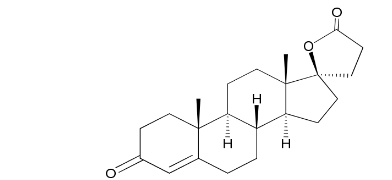
Spironolactone EP impurity C
| Product Name | Spironolactone EP impurity C |
|---|---|
| Alternate Names | Spironolactone Impurities, Impurities of Spironolactone |
| CAT No. | CS-O-10705 |
| CAS No. | 976-70-5 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 342.47 g/mol |
| Mol. For. | C₂₂H₃₀O₃ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Spironolactone |
| Purity | 98.97 |
| Therapeutic | Anti-Hypertensives |
| Smileys | CC12CCC(=O)C=C1CCC3C2CCC4(C3CCC45CCC(=O)O5)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Spironolactone EP impurity C is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for the analysis and identification of impurities in Spironolactone, a synthetic steroid that is used as a diuretic medication. Spironolactone EP impurity C is also known by its chemical name 3-(3-hydroxy-4-methoxyphenyl)-3-oxo-2-phenylpropanoic acid.
The chemical structure of Spironolactone EP impurity C consists of a benzene ring with a hydroxyl and methoxy group attached to it, along with a ketone and carboxylic acid functional group. It is a white to off-white crystalline powder that is sparingly soluble in water, but soluble in organic solvents such as methanol and acetonitrile.
As an impurity, Spironolactone EP impurity C can affect the purity, potency, and safety of Spironolactone. Therefore, it is important for pharmaceutical manufacturers to accurately identify and quantify this impurity in their products. This can be accomplished through the use of analytical techniques such as high-performance liquid chromatography (HPLC) and mass spectrometry (MS).
In conclusion, Spironolactone EP impurity C is a critical reference standard for the pharmaceutical industry to ensure the quality and safety of Spironolactone. Its chemical properties and analytical characteristics make it an important tool in the development and quality control of pharmaceuticals.
Get an Instant Quote
Related Compounds
Spironolactone Epimer Mixture | Spironolactone impurity H | Spironolactone EP impurity A | Spironolactone EP impurity G | Spironolactone impurity D | Spironolactone EP impurity B | Spironolactone acid | Spironolactone EP impurity E | Spironolactone Impurity 2 | Spironolactone EP impurity I |