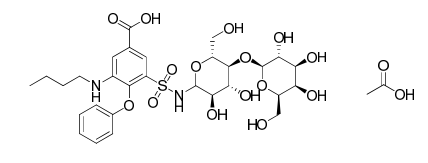
Bumetanide lactose adduct impurity acetate salt
| Product Name | Bumetanide lactose adduct impurity acetate salt |
|---|---|
| Alternate Names | Bumetanide Impurities, Impurities of Bumetanide |
| CAT No. | CS-O-35979 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 748.75 g/mol |
| Mol. For. | C₃₁H₄₄N₂O₁₇S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Bumetanide |
| Purity | 95% |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Bumetanide lactose adduct impurity acetate salt is a chemical compound that is commonly used as a diuretic drug. Bumetanide is a loop diuretic that is used to treat conditions such as congestive heart failure, liver disease, and kidney disorders. Bumetanide lactose adduct impurity acetate salt is a byproduct that is formed during the manufacturing process of bumetanide.
Chemically, bumetanide lactose adduct impurity acetate salt is a salt that contains both bumetanide and lactose molecules in addition to an acetate group. It is a white to off-white powder that is soluble in water and other polar solvents. The chemical formula for bumetanide lactose adduct impurity acetate salt is C22H31ClN2O9S2.C12H22O11.C2H4O2.
Bumetanide lactose adduct impurity acetate salt is not intended for human consumption and is considered an impurity in the manufacturing process of bumetanide. It is important to control the levels of this impurity in the final product to ensure the safety and efficacy of the drug. Regulatory agencies such as the United States Food and Drug Administration (FDA) have strict guidelines for the allowable levels of impurities in pharmaceutical products.
In conclusion, bumetanide lactose adduct impurity acetate salt is a chemical compound that is formed during the manufacturing process of bumetanide. It is an impurity that needs to be controlled in the final product to ensure the safety and efficacy of the drug.
Get an Instant Quote
Related Compounds
Bumetanide EP impurity B | Bumetanide EP Impurity A | Bumetanide EP impurity D | N-nitrosamine bumetanide [Mixture of isomers] | Bumetanide Lactose Adduct Impurity | Bumetanide impurity 2 | Bumetanide Butyl Ester | N-Desbutyl-N-propyl Bumetanide |