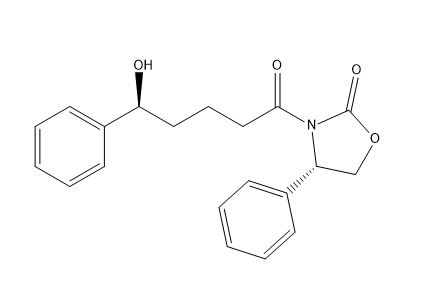
Ezetimibe Dides fluoro impurity
| Product Name | Ezetimibe Dides fluoro impurity |
|---|---|
| Alternate Names | Ezetimibe Impurities, Impurities of Ezetimibe |
| CAT No. | CS-O-15762 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 339.39 g/mol |
| Mol. For. | C₂₀H₂₁NO₄ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ezetimibe |
| Therapeutic | Anti-Hyperlipidemics |
| Smileys | O[C@H](C1=CC=CC=C1)CCCC(N2[C@@H](C3=CC=CC=C3)COC2=O)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ezetimibe Dides fluoro impurity is a chemical compound that is commonly used in the manufacturing of pharmaceutical products. This compound is an impurity that is found in the active ingredient, Ezetimibe Dides, which is used to treat high levels of cholesterol in the body. The fluoro impurity is a byproduct of the synthesis process of Ezetimibe Dides and is formed due to the incorporation of a fluorine atom into the molecule.
The usage of Ezetimibe Dides fluoro impurity is mainly in the pharmaceutical industry, where it is used as a reference standard in the development and quality control of Ezetimibe Dides-based products. It is an essential impurity that is monitored during the manufacturing process of Ezetimibe Dides to ensure that it is within acceptable limits.
Chemically, Ezetimibe Dides fluoro impurity is a derivative of Ezetimibe Dides, with the addition of a fluorine atom. It has a molecular weight of 409.43 g/mol, and its chemical formula is C24H22FNO3. The compound is a white to off-white powder that is soluble in organic solvents such as methanol and ethanol.
In conclusion, Ezetimibe Dides fluoro impurity is a crucial compound in the pharmaceutical industry, where it is used as a reference standard in the development and quality control of Ezetimibe Dides-based products. Its chemical formula and properties make it an essential impurity that is monitored during the manufacturing process of Ezetimibe Dides to ensure that it is within acceptable limits.
Get an Instant Quote
Related Compounds
Ezetimibe Related Impurity 7 | Ezetimibe Impurity 15 | m-Fluoroaniline isomer of Ezetimibe | O-Fluorobenzene isomer of Ezetimibe | Ezetimibe Triol Impurity | Ezetimibe Trihydroxy Impurity | Ezetimibe Lactone Impurity | Ezetimibe Impurity 10 | Ezetimibe 2-Fluoro impurity | Ezetimibe Benzyl Impurity (MBZT-2) | ent-Ezetimibe | RSR Ezetimibe | Ezetimibe Benzyl Diol Impurity | Ezetimibe Impurity B | Ezetimibe O-trimethylsilyl O-benzyl Impurity | Ezetimibe impurity (3-[5-(4-Fluoro-phenyl)-5-(R)-hydroxy-pentanoyl]-4-(S)-phenyl-oxazolidin-2-one) | Benzyl Ezetimibe ether | Ezetimibe Impurity 17 | Ezetimibe Diol Impurity | RRR-Ezetimibe+SSS-Ezetimibe (Diastereomer mixture) | Ezetimibe Impurity 11 | Ezetimibe Impurity C | Ezetimibe Impurity 18 | Ezetimibe Deprotected Impurity | Ezetimibe Desfluoro impurity | Ezetimibe Impurity 1 ((3'S,3R,4S)-Desfluoro Ezetimibe) | Ezetimibe (3R,4R,3'R)-Isomer | Ezetimibe Lactam Cleaved Alcohol | Ezetimibe (3S,4S,3'R)-Isomer | N-Nitroso Ezetimibe Impurity | O-Fluoroaniline isomer of Ezetimibe | Ezetimibe Diacid | Ezetimibe tetrahydropyran analog | Ezetimibe Didesfluro impurity | Ezetimibe ring open impurity | Ezetimibe Ring-opening Dehydrate Impurity | Benzylated Ezetimibe | Ezetimibe Impurity 25 | Ezetimibe Azetidinone Ring opened impurity | SSR-Ezetimibe | Ezetimibe 2-Fluoro Hydroxy impurity | Methyl 5-(4-Fluorophenyl)-(5S)-hydroxypentanoate |